Neuroanatomy: A Deeper Dive
An elaboration on Chapter 5: Macroscopic Organization of the Nervous System
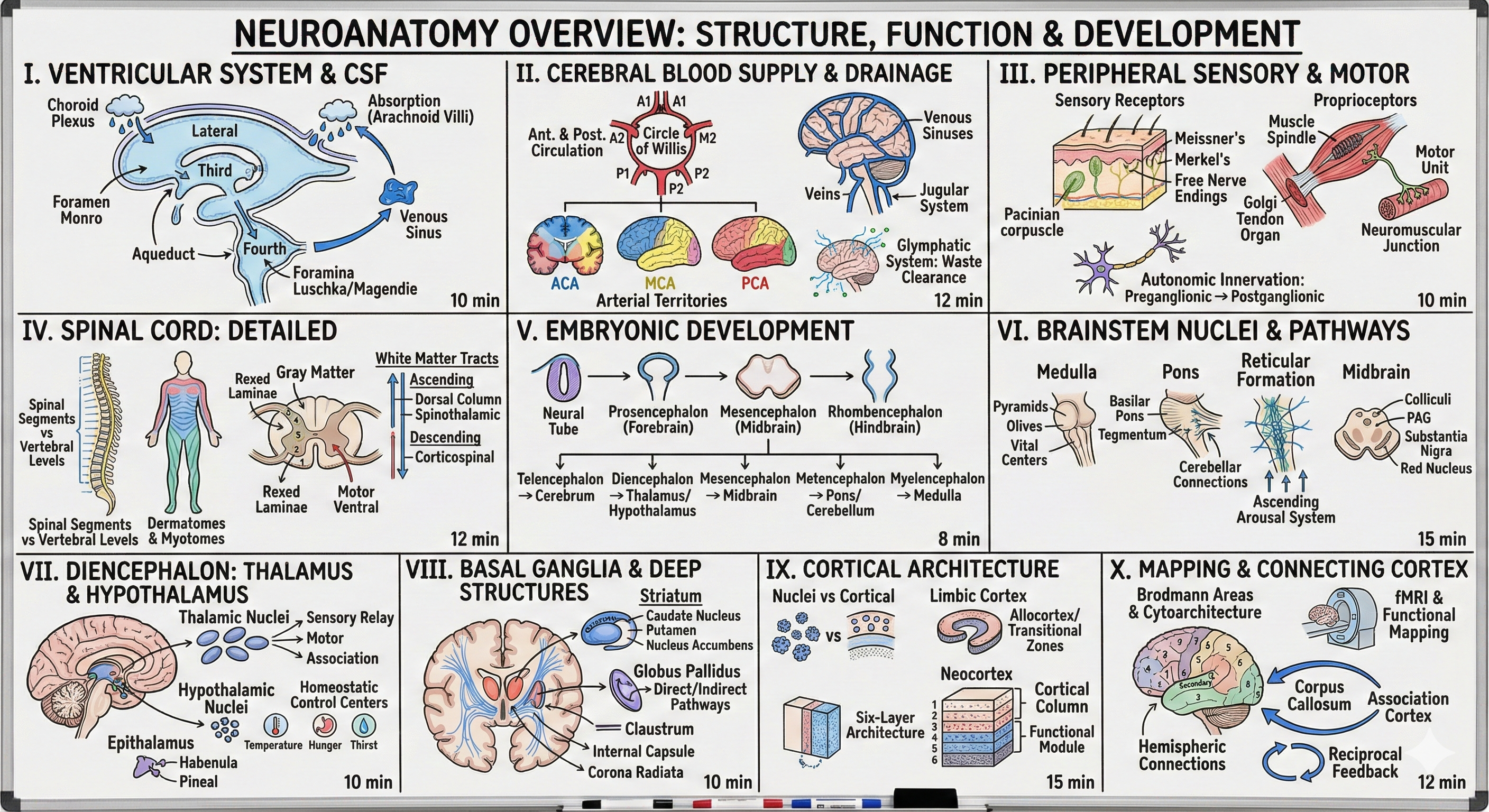
• Four ventricles and their connections
• Choroid plexus and CSF production
• CSF flow pathway and absorption
II. Cerebral Blood Supply and Drainage (12 min)
• Circle of Willis: anterior and posterior circulation
• Cerebral arteries and their territories
• Venous drainage: sinuses and jugular system
• Glymphatic system: the brain's waste clearance
III. Peripheral Sensory and Motor Systems (10 min)
• Cutaneous receptors and their specializations
• Proprioceptors: muscle spindles and Golgi tendon organs
• Neuromuscular junction and motor units
• Autonomic innervation: preganglionic and postganglionic neurons
IV. Spinal Cord: Detailed Organization (12 min)
• Spinal segments vs vertebral levels
• Dermatomes and myotomes: clinical significance
• Gray matter: Rexed laminae and functional zones
• White matter tracts: ascending and descending pathways
V. Embryonic Brain Development (8 min)
• Neural tube to brain vesicles
• Three primary vesicles: prosencephalon, mesencephalon, rhombencephalon
• Five secondary vesicles and adult derivatives
VI. Brainstem Nuclei and Pathways (15 min)
• Medulla: pyramids, olives, and vital centers
• Pons: basilar pons, tegmentum, and cerebellar connections
• Reticular formation and ascending arousal system
• Midbrain: colliculi, PAG, substantia nigra, red nucleus
VII. Diencephalon: Thalamic and Hypothalamic Nuclei (10 min)
• Thalamic nuclei: sensory relay, motor, and association
• Hypothalamic nuclei: homeostatic control centers
• Habenula and pineal: the epithalamus
VIII. Basal Ganglia and Deep Structures (10 min)
• Striatum: caudate, putamen, nucleus accumbens
• Globus pallidus and direct/indirect pathways
• Claustrum: the mysterious sheet
• Internal capsule and corona radiata
IX. Cortical Architecture (15 min)
• Nuclei vs cortical organization
• Limbic cortex: allocortex and transitional zones
• Neocortex: six-layer architecture
• Cortical columns: functional modules
X. Mapping and Connecting the Cortex (12 min)
• Brodmann areas and cytoarchitecture
• fMRI and modern functional mapping
• Primary, secondary, and association cortex
• Corpus callosum and hemispheric connections
• Reciprocal feedback at every level
This chapter provides a detailed anatomical elaboration of the nervous system organization introduced in Chapter 5. We begin with the ventricular system that bathes your brain in cerebrospinal fluid and the arterial circle that ensures continuous blood supply—including the recently discovered glymphatic system that cleans your brain while you sleep. We then explore the peripheral receptors that detect touch, position, and movement, before diving into the precise segmental organization of the spinal cord with its clinical dermatomes and white matter highways. Embryonic development reveals how three simple vesicles become the five major brain divisions, setting the stage for our tour of brainstem nuclei—from the medulla's vital centers to the midbrain's reward circuits. The diencephalon's thalamic relay stations and hypothalamic control centers give way to the basal ganglia's movement selection circuits and the cortex's layered architecture. Finally, we map the cortex from Brodmann's cytoarchitectonic areas to modern fMRI discoveries, always returning to a central theme: every structure connects reciprocally with others. The brain is not a one-way hierarchy but a dynamic network of feedback loops at every level.
Part I: The Ventricular System
Four Ventricles and Their Connections
[VIEW IMAGES: Ventricular system showing lateral, third, and fourth ventricles]
Deep within your brain lies an ancient plumbing system—a network of fluid-filled cavities that traces its origins to the hollow tube from which your entire nervous system developed. The ventricular system consists of four interconnected chambers that house and circulate cerebrospinal fluid throughout your central nervous system. Two large C-shaped lateral ventricles occupy the cerebral hemispheres—one in each side of your brain—curving through the frontal, parietal, occipital, and temporal lobes like a pair of ram's horns. Sandwiched between the two thalami at the brain's center sits the narrow, slit-like third ventricle, shaped more like a flattened vertical coin than a balloon. Below and behind this, nestled between the brainstem and cerebellum, the tent-shaped fourth ventricle completes the system before the central canal continues down through the spinal cord.
Each lateral ventricle mirrors the complex geography of the cerebral hemisphere it occupies. The frontal horn extends into the frontal lobe, giving way to the body of the ventricle as you move posteriorly through the parietal region. At the atrium—also called the trigone—the ventricular space widens at the crossroads where three directions diverge: the occipital horn reaches backward into the occipital lobe, the temporal horn curves downward and forward into the temporal lobe like a hook, and the body continues from above. This shape perfectly reflects how the cerebral hemispheres grew and folded during development. The lateral ventricles communicate with the third ventricle through a pair of small openings called the interventricular foramina of Monro—narrow passages that occasionally become blocked, trapping fluid in one lateral ventricle and causing asymmetric swelling.
The third ventricle sits in the precise midline of the brain, bounded on each side by the medial walls of the two thalami, below by the hypothalamus, and anteriorly by the delicate lamina terminalis—a thin membrane marking the original front end of the embryonic neural tube. From its posterior end, the fluid pathway narrows dramatically as it enters the cerebral aqueduct of Sylvius, a channel no wider than a pencil lead that tunnels through the midbrain. This aqueduct represents the most vulnerable chokepoint in the entire CSF pathway; even slight narrowing from tumor, infection, or developmental abnormality can obstruct flow and cause the ventricles above to balloon dangerously—a condition called obstructive hydrocephalus.
The fourth ventricle opens like a tent beneath the cerebellum, with the pons and medulla forming its floor—a region called the rhomboid fossa because of its diamond shape when viewed from above. This floor contains crucial brainstem nuclei, including those controlling facial movement, hearing, balance, and vital functions. Cerebrospinal fluid escapes the ventricular system through three apertures in the fourth ventricle's walls: the median aperture of Magendie opens at the back, while two lateral apertures of Luschka allow fluid to flow into the subarachnoid space surrounding the brain and spinal cord. Once outside the ventricles, CSF circulates around the entire central nervous system, cushioning it, feeding it, and eventually draining back into the bloodstream.
CSF Production, Circulation, and Absorption
[VIEW IMAGES: Choroid plexus structure and CSF circulation pathway]
Your brain manufactures its own bathwater at an astonishing rate. The choroid plexus—specialized secretory tissue found in each ventricle—produces approximately 500 milliliters of cerebrospinal fluid every day, meaning the entire CSF volume of about 150 milliliters turns over three to four times daily. These factories consist of ependymal cells modified into secretory epithelium, their surfaces covered with microvilli to increase absorption area, their connections sealed with tight junctions to prevent uncontrolled leakage. Beneath this epithelium lies a core of highly vascularized connective tissue from which blood plasma is filtered. The choroid plexus cells don't merely filter passively—they actively transport specific ions across their membranes, and water follows osmotically, creating a fluid with a carefully controlled composition distinct from blood plasma.
The resulting cerebrospinal fluid is essentially a clarified, modified ultrafiltrate of blood, but the differences matter enormously for neural function. CSF contains far less protein than plasma—about 0.3% compared to plasma's 7%—creating a clear, water-like fluid that won't clog the narrow passages it must traverse. Potassium concentration is kept lower than in blood, glucose slightly lower, while sodium and chloride are maintained at precise levels. Most strikingly, CSF normally contains almost no cells—fewer than five lymphocytes per microliter, compared to the thousands of cells per microliter in blood. This near-sterility makes CSF analysis invaluable for diagnosis: finding bacteria, cancer cells, or elevated white cell counts immediately signals infection or disease.
From its birthplace in the choroid plexus, CSF embarks on a one-way journey through an elegant circulatory pathway. Beginning in the lateral ventricles, fluid flows through the interventricular foramina into the third ventricle, then squeezes through the narrow cerebral aqueduct into the fourth ventricle. From there, it escapes into the subarachnoid space through the apertures of Magendie and Luschka, spreading around the brain and spinal cord in the space between the arachnoid and pia mater. This fluid bathes every surface of the CNS, filling the sulci between gyri, surrounding the cranial nerves at their exit points, and extending down around the spinal cord to the lumbar cistern below where the cord ends. Finally, CSF drains back into the venous blood through arachnoid granulations—mushroom-shaped protrusions that push through the dura into the dural venous sinuses, acting as one-way valves that open when CSF pressure exceeds venous pressure.
When this elegant circulation fails, the consequences can be devastating. Hydrocephalus—literally "water on the brain"—occurs when CSF accumulates faster than it drains, causing ventricular enlargement and increased intracranial pressure. Obstructive or non-communicating hydrocephalus develops when blockage prevents CSF from flowing between ventricles, most commonly at the narrow cerebral aqueduct. Communicating hydrocephalus occurs when fluid can circulate but cannot be absorbed at the arachnoid granulations—perhaps due to scarring from prior meningitis or subarachnoid hemorrhage. In infants whose skull sutures haven't yet fused, hydrocephalus causes dramatic head enlargement; in adults with rigid skulls, pressure builds internally, compressing brain tissue and causing headaches, cognitive decline, and eventually death if untreated. The standard treatment involves placing a ventriculoperitoneal shunt—a thin tube that diverts excess CSF from the ventricles to the abdominal cavity, where it absorbs harmlessly into the body.
CSF provides cushioning, but the brain's voracious metabolic demands require something more: a constant, abundant blood supply. The arterial system that delivers this blood has its own elegant redundancies and vulnerabilities.
Part II: Cerebral Blood Supply
The Circle of Willis
[VIEW IMAGES: Circle of Willis showing anterior and posterior circulation]
Your brain's hunger for blood is voracious and unforgiving—though it represents only 2% of body weight, it demands 15-20% of cardiac output at all times. To meet this demand while protecting against catastrophic failure, evolution engineered a remarkable redundancy: the circle of Willis, an arterial ring at the brain's base where two independent supply systems converge and interconnect. The anterior circulation arrives via the internal carotid arteries—paired vessels that climb through bony channels in the skull base called the carotid canals—while the posterior circulation ascends via the vertebral arteries, which thread through holes in the cervical vertebrae before entering the skull through the foramen magnum. If one pathway fails, blood can theoretically reroute through the other, though this elegant backup system works perfectly in only about a quarter of the population.
The internal carotid arteries bifurcate to form the workhorses of cerebral circulation. The anterior cerebral arteries curl forward and upward between the hemispheres, supplying the medial surfaces of the frontal and parietal lobes—including crucially, the leg areas of the motor and sensory cortex that wrap over onto the medial surface. Stroke here produces leg weakness and sensory loss while sparing the arm and face. The middle cerebral arteries—the largest branches—sweep laterally into the Sylvian fissure, supplying the vast lateral surface of each hemisphere: the motor and sensory cortex for face and arm, the language areas in the dominant hemisphere, and the lenticulostriate branches that penetrate deeply to feed the basal ganglia and internal capsule. The middle cerebral artery is the most common site of stroke, and its occlusion produces the classic picture of contralateral face and arm weakness with speech disturbance. Meanwhile, the ophthalmic arteries branch off to supply the retina—their blockage causes sudden, painless blindness in one eye.
The posterior circulation begins when the two vertebral arteries merge at the pontomedullary junction to form the single basilar artery, which runs along the front surface of the pons. Before merging, each vertebral artery gives off a posterior inferior cerebellar artery (PICA), whose occlusion produces the dramatic lateral medullary (Wallenberg) syndrome. The basilar artery sprouts the anterior inferior cerebellar and superior cerebellar arteries before terminating as the paired posterior cerebral arteries, which wrap around the midbrain to supply the occipital lobes and inferior temporal lobes. Posterior cerebral occlusion produces isolated visual field defects—the patient may be cortically blind while their eyes remain perfectly normal.
The circle is completed by communicating arteries that bridge the two systems. The anterior communicating artery connects the two anterior cerebrals across the midline—this tiny vessel is the most common site of cerebral aneurysm. The posterior communicating arteries connect each internal carotid to the posterior cerebral artery on its side, bridging anterior and posterior circulations. Yet despite this apparent redundancy, a complete and symmetric circle exists in only about 25% of people—most have hypoplastic or absent communicating segments that limit collateral flow. This anatomical variability explains why some patients survive carotid occlusion with minimal symptoms while others suffer massive strokes: the effectiveness of the backup depends on the individual's vascular anatomy, developed before birth but only tested when disaster strikes.
Venous Drainage: Dural Sinuses
[VIEW IMAGES: Dural venous sinuses and cerebral venous drainage]
Unlike the rest of your body, where arteries and veins travel together in neurovascular bundles, the brain's venous system follows its own independent geography—and for good reason. Cerebral veins are thin-walled, valveless vessels that drain into the dural venous sinuses: rigid channels formed between the two layers of the dura mater where it folds upon itself. Because these sinuses are enclosed within tough, fibrous dura, they cannot collapse even when intracranial pressure fluctuates wildly, ensuring continuous venous drainage regardless of head position, coughing, or straining. This engineering solution allows the brain's venous system to work passively, driven purely by pressure gradients rather than the muscular pumping and valves that move blood through limb veins.
The largest sinus, the superior sagittal sinus, runs along the top of the falx cerebri from the frontal bone to the back of the skull, growing progressively larger as it receives cortical veins draining the brain's convex surface. Its walls are studded with arachnoid granulations—the mushroom-like protrusions through which CSF drains into the venous blood. Below and parallel to this runs the inferior sagittal sinus along the free edge of the falx, which meets the great vein of Galen—a massive vessel draining the brain's deep structures—to form the straight sinus. These three major channels converge at the confluence of sinuses, a blood-filled crossroads at the internal occipital protuberance where the falx meets the tentorium.
From the confluence, blood flows laterally through the paired transverse sinuses, which groove the inner surface of the occipital bone as they curve toward the temporal region. Each transverse sinus turns sharply downward to become the sigmoid sinus—so named for its S-shaped course—which exits the skull through the jugular foramen as the internal jugular vein. The transverse sinuses are often asymmetric: one side (usually the right) is dominant and drains most of the brain, while the other may be hypoplastic. Thrombosis of a dural sinus—from dehydration, infection, or hypercoagulable states—can produce devastating venous infarction as blood backs up into the brain, causing hemorrhage, seizures, and raised intracranial pressure.
The Glymphatic System: Brain Waste Clearance
[VIEW IMAGES: Glymphatic system showing CSF flow and waste clearance]
For over a century, neuroscientists puzzled over a peculiar absence: the brain lacks conventional lymphatic vessels within its tissue, yet somehow manages to clear metabolic waste without the lymphatic drainage that other organs depend upon. The answer, discovered only in 2012, is the glymphatic system—a brain-wide waste clearance pathway that operates through the paravascular spaces surrounding blood vessels. This glial-dependent lymphatic-like system uses cerebrospinal fluid as a cleaning fluid, flushing through the brain tissue to collect and remove metabolic debris. The discovery has revolutionized our understanding of why sleep matters and what happens when the brain can't take out its trash.
The mechanism is elegant in its simplicity. CSF flows into the brain along the paravascular spaces—also called Virchow-Robin spaces—that surround penetrating arteries. This influx is facilitated by aquaporin-4 (AQP4) water channels concentrated on the end-feet of astrocytes, the star-shaped glial cells that wrap around every blood vessel in the brain. As CSF percolates through the brain's interstitial spaces, it mixes with interstitial fluid and picks up metabolic waste products—including amyloid-beta and tau proteins, the very molecules that accumulate in Alzheimer's disease. The waste-laden fluid then drains along venous paravascular spaces, eventually reaching the recently discovered meningeal lymphatic vessels that carry it to cervical lymph nodes.
The most remarkable feature of the glymphatic system is its state dependence: during sleep, particularly during deep slow-wave NREM sleep, glymphatic flow increases by approximately 60% compared to waking. This dramatic enhancement occurs because neurons physically shrink during sleep, expanding the extracellular space by about 60% and dramatically reducing resistance to fluid flow—imagine the difference between forcing water through packed gravel versus loose pebbles. Norepinephrine levels, which are high during waking and suppress glymphatic flow, drop precipitously during sleep, releasing the brake on this cleaning mechanism. The implications are profound: chronic sleep deprivation doesn't just make you tired, it impairs your brain's ability to clear toxic metabolic waste, potentially accelerating neurodegeneration. This may explain why poor sleep is consistently associated with increased Alzheimer's risk, and why a single night of sleep deprivation increases brain amyloid-beta levels.
The central nervous system may be protected and nourished, but it accomplishes nothing without connections to the outside world. Information must flow in from sensors throughout the body, and commands must flow out to muscles that move the skeleton. We turn now to the peripheral components that interface brain with body.
Part III: Peripheral Sensory and Motor Components
Cutaneous Sensory Receptors
[VIEW IMAGES: Cutaneous receptors showing different receptor types]
Your skin is not merely a covering—it is an immense sensory organ studded with millions of specialized receptors, each exquisitely tuned to detect specific features of the physical world. These receptors fall into several functional categories, with mechanoreceptors forming the backbone of tactile sensation. Four main mechanoreceptor types tile the skin in a precise mosaic, each distinguished by its adaptation properties and receptive field size—characteristics that determine what aspect of touch it encodes. Together, these receptors enable the remarkable discriminative abilities of human touch: the capacity to read Braille, detect the edge of a page, or identify a familiar object by feel alone.
Meissner's corpuscles cluster in the dermal papillae just beneath the epidermis, particularly dense in the fingertips and lips where fine tactile discrimination matters most. These rapidly adapting receptors with small receptive fields respond brilliantly to light touch and flutter, encoding the fine textures and edges of objects as your fingers move across them. In contrast, Pacinian corpuscles lie deeper in the dermis and subcutaneous tissue, their onion-like layered capsule perfectly engineered to filter out sustained pressure while transmitting high-frequency vibration in the 200-300 Hz range—the frequencies produced when your hand slides across textured surfaces or when tools vibrate in your grip.
Where Meissner's and Pacinian corpuscles adapt rapidly and signal change, Merkel discs and Ruffini endings adapt slowly and signal sustained states. Merkel discs, with their small receptive fields, resolve fine spatial detail—they're what allow you to feel the embossed letters on a credit card without looking. Ruffini endings, with larger receptive fields, detect skin stretch, signaling joint position and the deformation of skin during grip. Beyond mechanoreceptors, the skin contains separate populations of thermoreceptors for warm and cold, plus nociceptors—the pain receptors that detect tissue damage through free nerve endings distributed throughout the skin. This sensory array transforms your skin into an exquisitely sensitive interface with the physical world.
Proprioceptors: Position and Movement
[VIEW IMAGES: Muscle spindle and Golgi tendon organ structure]
Close your eyes and touch your nose—the ease with which you accomplish this reveals the power of proprioception, your sense of body position and movement. This sixth sense depends on specialized receptors embedded within muscles, tendons, and joints, continuously reporting to your central nervous system the precise configuration and motion of every limb. Without proprioception, movement becomes halting and clumsy, requiring constant visual monitoring; patients who lose this sense describe their limbs as foreign, present but disconnected from the self.
Muscle spindles are encapsulated receptors scattered throughout the belly of every skeletal muscle, lying parallel to the regular muscle fibers that generate force. Each spindle contains specialized intrafusal muscle fibers—thin fibers distinct from the force-generating extrafusal fibers surrounding them—with sensory nerve endings wrapped around their central region. When the muscle stretches, intrafusal fibers stretch with it, deforming the sensory terminals and triggering action potentials. Two types of sensory afferents convey complementary information: large-diameter Ia afferents from primary endings respond to both the length and velocity of stretch, while smaller Group II afferents from secondary endings encode primarily static length. This dual-channel design provides the nervous system with a complete picture of muscle state.
While spindles measure muscle length, Golgi tendon organs measure muscle force. Located at the junction between muscle and tendon, these receptors consist of collagen fibers interwoven with the terminals of Ib sensory afferents. When the muscle contracts and pulls on the tendon, the collagen fibers compress against the nerve terminals, generating signals proportional to the force of contraction. Together with joint receptors in capsules and ligaments that signal joint angle—particularly at the extremes of range—these proprioceptors provide the constant background information that makes coordinated movement possible. Every reach, every step, every gesture depends on this silent conversation between muscle and brain.
The Neuromuscular Junction
[VIEW IMAGES: Neuromuscular junction showing motor end plate]
Every voluntary movement you make—from typing these words to running a marathon—ultimately depends on a specialized synapse where nerve meets muscle: the neuromuscular junction. Here, alpha motor neurons in the spinal cord's ventral horn send their axons out through peripheral nerves to contact skeletal muscle fibers. Each motor neuron branches to innervate multiple muscle fibers—from just a handful in eye muscles requiring fine control to over a thousand in large leg muscles built for power—and one motor neuron plus all the muscle fibers it commands constitutes a motor unit, the fundamental unit of motor control.
At each junction, the motor neuron terminal releases acetylcholine onto a specialized region of the muscle membrane called the motor end plate. This postsynaptic membrane is densely packed with nicotinic acetylcholine receptors—ligand-gated ion channels that open when acetylcholine binds, allowing sodium to rush into the muscle cell and triggering a local depolarization called the end-plate potential. Unlike synapses in the brain where a single input produces only small postsynaptic changes, the neuromuscular junction is designed for reliable, one-to-one transmission: every action potential in the motor neuron produces an action potential in the muscle fiber, which then propagates along the fiber membrane and deep into the cell through T-tubules, ultimately triggering calcium release from the sarcoplasmic reticulum and muscle contraction.
Alongside alpha motor neurons, smaller gamma motor neurons innervate the intrafusal fibers within muscle spindles—not to generate force, but to maintain spindle sensitivity across all muscle lengths. When a muscle shortens, intrafusal fibers would go slack and stop signaling were it not for gamma motor neurons simultaneously contracting their ends, keeping the central sensory region taut. This alpha-gamma coactivation ensures that proprioceptive feedback remains reliable whether your biceps is stretched long or contracted short. The neuromuscular junction thus sits at the critical output node of the motor system, translating neural signals into the mechanical force that moves your skeleton through the world.
Autonomic Innervation
[VIEW IMAGES: Autonomic innervation showing two-neuron chains]
While somatic motor neurons project directly from spinal cord to muscle in a single-neuron pathway, the autonomic nervous system employs a mandatory two-neuron chain to reach its targets. Preganglionic neurons with cell bodies in the central nervous system send relatively thin, lightly myelinated axons to peripheral ganglia, where they synapse on postganglionic neurons that complete the pathway to target organs. This relay architecture serves both anatomical and physiological purposes: it allows divergence and convergence of signals in the ganglia, and it separates the protected preganglionic neurons within the CNS from the more vulnerable postganglionic neurons in the periphery.
The sympathetic division—orchestrating the "fight or flight" response—arises from the thoracolumbar spinal cord segments T1 through L2. Preganglionic sympathetic neurons send their axons through the ventral roots into the spinal nerves, then immediately branch into the sympathetic chain—a paired string of ganglia running alongside the vertebral column—via white rami communicantes. Within this chain, preganglionic fibers may synapse at the level they enter, ascend or descend to synapse at other chain ganglia, or pass through entirely to reach prevertebral ganglia closer to their target organs—the celiac, superior mesenteric, and inferior mesenteric ganglia that innervate abdominal viscera. Postganglionic sympathetic neurons use norepinephrine as their neurotransmitter at most targets, with the notable exception of sweat glands, which receive sympathetic innervation but respond to acetylcholine.
The parasympathetic division—promoting "rest and digest" functions—has a craniosacral origin, arising from brainstem nuclei and sacral spinal segments S2-S4. Cranial parasympathetic outflow travels with four cranial nerves: the oculomotor (III) to the pupillary constrictor and ciliary muscle, the facial (VII) to lacrimal and salivary glands, the glossopharyngeal (IX) to the parotid gland, and most extensively, the vagus nerve (X), which provides parasympathetic innervation to the heart, lungs, and nearly all abdominal viscera as far as the splenic flexure of the colon. Unlike the sympathetic division's short preganglionic and long postganglionic fibers, parasympathetic preganglionic fibers are long, synapsing in ganglia located near or within the walls of target organs. Both preganglionic and postganglionic parasympathetic neurons use acetylcholine as their neurotransmitter.
All these peripheral sensory and motor pathways converge on the spinal cord—the first processing station where reflexes are organized and information is sorted for its journey to or from the brain. The cord's precise segmental organization provides a framework for understanding both normal function and the clinical patterns of spinal pathology.
Part IV: Spinal Cord Organization
Spinal Segments and Vertebral Levels
[VIEW IMAGES: Spinal cord segments and their relationship to vertebral levels]
The spinal cord is organized into 31 segments, each defined by a pair of spinal nerves that emerge from its sides—eight cervical segments (C1-C8), twelve thoracic (T1-T12), five lumbar (L1-L5), five sacral (S1-S5), and one coccygeal. But here lies a crucial anatomical fact that trips up students and clinicians alike: the spinal cord is shorter than the vertebral column that houses it. During fetal development, the spinal cord initially extends the full length of the vertebral canal, but the vertebral column grows faster, so that by adulthood the cord ends at approximately the L1-L2 vertebral level in a tapered terminus called the conus medullaris. Below this point, the vertebral canal contains no spinal cord—only the nerve roots of the lumbar and sacral segments streaming downward to exit at their appropriate levels, forming the cauda equina, so named for its resemblance to a horse's tail.
This anatomical relationship between cord segments and vertebral levels has critical clinical implications. In the cervical region, there are seven vertebrae but eight spinal nerves because C1 through C7 exit above their corresponding vertebrae, while C8 exits between C7 and T1. From T1 downward, each nerve exits below its corresponding vertebra—the T1 nerve exits below the T1 vertebra, and so on. But because the cord ends at L1-L2, the lumbar and sacral cord segments are actually located within the thoracolumbar vertebral column: approximately T10 vertebral level corresponds to the L1-L2 spinal segments, while the conus medullaris containing the sacral segments lies behind the L1 vertebra. A penetrating injury at the T12 vertebral level therefore damages not thoracic but lumbar spinal cord, producing weakness in the legs rather than the trunk.
The spinal cord is not uniform in thickness—two prominent enlargements mark the regions that innervate the limbs, where extra neurons are needed for the complex motor control and sensory processing that limbs require. The cervical enlargement spans approximately C3 through T1, supplying the brachial plexus and upper extremities; the lumbar enlargement from L1 through S2 supplies the lumbosacral plexus and lower extremities. Between and beyond these enlargements, the cord is narrower where it serves only the trunk and viscera. This simple observation explains why complete spinal cord injury at different levels produces different patterns of paralysis: cervical injuries cause quadriplegia affecting all four limbs, while thoracic injuries cause paraplegia with preserved arm function.
Dermatomes and Myotomes
[VIEW IMAGES: Dermatome map showing sensory distribution]
The segmental organization of the spinal cord is written onto the surface of your body as dermatomes—strips of skin each supplied by sensory fibers from a single spinal nerve. When a patient reports numbness in a dermatomal pattern, the clinician can immediately pinpoint which spinal segment is likely damaged. The dermatome map wraps around the body in a logical pattern reflecting our embryonic development: C4 covers the shoulders in a cape-like distribution, C6 includes the thumb, C8 reaches the little finger, T4 crosses the nipple line, T10 encircles the umbilicus, L1 covers the inguinal region, L4 extends down the medial leg to the ankle, S1 supplies the lateral foot, and S2-S4 form the saddle area of the perineum. Because adjacent dermatomes overlap substantially, complete anesthesia in a dermatomal distribution typically requires damage to at least two consecutive segments.
Just as dermatomes map sensation, myotomes map motor control—each spinal segment innervates a specific group of muscles. Testing myotomes helps localize lesions when weakness is present. A patient with weakness of shoulder abduction (C5) and elbow flexion (C6) but normal elbow extension (C7) likely has a lesion at the C5-C6 level. Key myotomal landmarks include C5 for deltoid and biceps, C6 for wrist extensors, C7 for triceps and wrist flexors, C8 for finger flexors, T1 for hand intrinsics, L2 for hip flexors, L3 for knee extensors, L4 for ankle dorsiflexors, L5 for great toe extensors, and S1 for ankle plantarflexors. The combination of sensory dermatomal and motor myotomal examination, together with reflex testing (which depends on intact segmental arcs), allows remarkably precise localization of spinal pathology.
Gray Matter: Rexed Laminae
[VIEW IMAGES: Spinal cord gray matter showing Rexed laminae]
In cross-section, the spinal cord reveals its fundamental design: a butterfly-shaped core of gray matter surrounded by white matter highways. The gray matter—containing neuron cell bodies, dendrites, and synapses—is organized not randomly but into ten distinct laminae described by Bror Rexed in 1952 and known as the Rexed laminae, numbered I through X from the dorsal to ventral surface. This laminar organization reflects function: the dorsal horn processes incoming sensory information, the ventral horn contains the motor neurons that drive movement, and the intermediate zone integrates and coordinates.
The dorsal horn, comprising laminae I through VI, is the sensory processing center. Lamina I, the thin marginal zone at the dorsal tip, receives pain and temperature information and sends projections into the spinothalamic tract. Just beneath it, lamina II—the substantia gelatinosa—is rich in enkephalins and substance P and serves as a crucial site for pain modulation; this is where descending analgesic systems and local interneurons can "gate" pain signals before they ascend to the brain. Laminae III and IV, collectively the nucleus proprius, process light touch. Laminae V and VI handle proprioceptive input, with Clarke's column in lamina VII (at C8-L3 levels) relaying unconscious proprioception to the cerebellum via the dorsal spinocerebellar tract.
The intermediate zone, lamina VII, contains interneurons that coordinate sensory-motor integration and, crucially at thoracolumbar levels T1-L2 and sacral levels S2-S4, houses the intermediolateral cell column—the preganglionic neurons of the autonomic nervous system. This lateral horn, visible as a small projection of gray matter between dorsal and ventral horns, contains the sympathetic preganglionic neurons (T1-L2) and sacral parasympathetic neurons (S2-S4) that control visceral function. The ventral horn, laminae VIII and IX, is the motor output zone. Lamina VIII contains interneurons that modulate motor neuron activity, while lamina IX houses the alpha and gamma motor neurons themselves—arranged somatotopically with medial motor neurons innervating axial muscles and lateral motor neurons innervating limb muscles. Surrounding the central canal, lamina X completes the scheme.
White Matter Tracts
[VIEW IMAGES: Spinal cord white matter tracts]
The white matter surrounding the spinal gray matter is organized into three columns or funiculi—dorsal, lateral, and ventral—each containing specific fiber bundles or tracts that carry information up to the brain or down from it. The ascending sensory tracts and descending motor tracts are precisely organized, their positions within the white matter so consistent that damage to specific cord regions produces predictable, localizable deficits. Understanding which tracts cross at which levels explains the peculiar dissociations that spinal cord lesions produce.
Two major ascending systems carry different sensory modalities. The dorsal columns—the fasciculus gracilis carrying information from the lower body and fasciculus cuneatus from the upper body—transmit fine touch, vibration sense, and conscious proprioception. These fibers ascend ipsilaterally all the way to the medulla before crossing, which means that damage to the dorsal columns produces sensory loss on the same side as the lesion. In contrast, the spinothalamic tracts in the anterolateral white matter carry pain, temperature, and crude touch—but these fibers cross within a segment or two of entering the cord, so spinothalamic damage produces sensory loss on the opposite side. The spinocerebellar tracts, both dorsal and ventral, carry unconscious proprioceptive information to the cerebellum for motor coordination.
The descending motor tracts follow equally precise organization. The lateral corticospinal tract, carrying voluntary motor commands from the motor cortex, crosses in the medulla at the pyramidal decussation before descending in the lateral funiculus—damage above the decussation produces contralateral weakness, while damage to the tract in the cord produces ipsilateral weakness. The smaller anterior corticospinal tract descends uncrossed and controls axial muscles, crossing at segmental levels. Additional descending tracts—the rubrospinal from the red nucleus (facilitating flexor movements), the vestibulospinal (maintaining balance and extensor tone), and the reticulospinal (controlling posture and locomotion)—modulate spinal motor output. The clinical syndrome of Brown-Séquard, caused by hemisection of the spinal cord, elegantly demonstrates these crossing patterns: patients show ipsilateral motor loss and proprioceptive loss (from damage to the already-crossed corticospinal tract and not-yet-crossed dorsal columns) combined with contralateral pain and temperature loss (from damage to the already-crossed spinothalamic tract).
The spinal cord's elegant organization—its segments, its laminae, its ascending and descending tracts—didn't emerge by accident. It reflects the developmental blueprint laid down in the earliest weeks of embryonic life. To understand why the adult brain is organized as it is, we must trace its origins back to the neural tube.
Part V: Embryonic Brain Development
From Neural Tube to Brain Vesicles
[VIEW IMAGES: Embryonic brain vesicle development]
Every human brain begins as a simple tube. During the third week of embryonic development, a process called neurulation transforms a flat plate of ectoderm into the neural tube—the primordium of the entire central nervous system. The tube's cavity will become the ventricular system; its walls will become the brain and spinal cord. By the end of the fourth week, the rostral end of this tube has already begun to expand, forming three primary brain vesicles: the prosencephalon (forebrain), mesencephalon (midbrain), and rhombencephalon (hindbrain). These Greek names—meaning "front brain," "middle brain," and "diamond-shaped brain"—persist in modern neuroanatomy because they describe real developmental and functional relationships.
By the fifth week, these three vesicles have subdivided into five secondary vesicles that correspond directly to the major divisions of the adult brain. The prosencephalon divides into the telencephalon ("end brain") anteriorly and the diencephalon ("between brain") posteriorly. The telencephalon will become the cerebral hemispheres, basal ganglia, and limbic structures—everything we associate with higher cognition—while its cavity expands into the lateral ventricles. The diencephalon will become the thalamus, hypothalamus, epithalamus, and subthalamus, with its cavity forming the narrow third ventricle. The mesencephalon remains undivided, becoming the adult midbrain—tectum, tegmentum, and cerebral peduncles—with its cavity narrowing into the cerebral aqueduct.
The rhombencephalon divides into the metencephalon ("after brain") rostrally and myelencephalon ("marrow brain") caudally. The metencephalon gives rise to the pons and cerebellum, while the myelencephalon becomes the medulla oblongata. Together, their cavities form the fourth ventricle, and together with the mesencephalon, they comprise the brainstem. Understanding this embryological progression does more than satisfy historical curiosity—it explains why certain structures are grouped together functionally and anatomically, why the ventricles have their peculiar shapes, and why developmental abnormalities produce the patterns they do. The brain's adult organization is a palimpsest written over the embryonic blueprint that every neuroscience student must learn to read.
With the embryonic framework in mind, we can now explore the adult brainstem in detail—the three segments that emerged from the mesencephalon, metencephalon, and myelencephalon, packed with nuclei that control everything from breathing to consciousness.
Part VI: Brainstem Nuclei and Pathways
Medulla Oblongata: Vital Centers
[VIEW IMAGES: Medulla anatomy showing pyramids, olives, and cranial nerve nuclei]
The medulla oblongata, the most caudal segment of the brainstem, contains the neural machinery that keeps you alive from heartbeat to heartbeat. Look at its ventral surface and you'll see paired ridges called the pyramids—so named for their shape—containing the massive descending corticospinal fibers that will control voluntary movement. At the pyramids' caudal end lies the pyramidal decussation, where 85% of these fibers cross to the opposite side, explaining why your right motor cortex controls your left hand. Lateral to the pyramids bulge the inferior olives—wrinkled structures resembling actual olives—whose neurons relay information to the cerebellum and play a crucial role in motor learning and error correction.
Deep within the medulla lie the vital centers that control the rhythms of life. The respiratory centers—a dorsal respiratory group driving inspiration and a ventral respiratory group coordinating expiration—generate the automatic breathing rhythm that continues whether you're awake or asleep. Nearby, cardiac centers balance cardioacceleratory and cardioinhibitory influences to fine-tune heart rate moment by moment. The vasomotor center adjusts vascular tone throughout the body, controlling blood pressure. Damage to the medulla is often fatal precisely because these centers are irreplaceable—there is no backup system for breathing or circulation.
Four cranial nerves have their nuclei within the medulla. The glossopharyngeal nerve (IX) controls swallowing and receives taste from the posterior tongue. The vagus nerve (X), the "wanderer," extends parasympathetic control to the heart, lungs, and most of the abdominal viscera. The accessory nerve (XI) turns your head (sternocleidomastoid) and shrugs your shoulders (trapezius). The hypoglossal nerve (XII) moves your tongue—damage causes it to deviate toward the lesion on protrusion. The medulla also contains the gracile and cuneate nuclei, where dorsal column sensory fibers synapse before crossing as the medial lemniscus on their way to the thalamus.
Pons and Cerebellum: Coordination
[VIEW IMAGES: Pons and cerebellum showing cerebellar peduncles]
Rising above the medulla, the pons—Latin for "bridge"—earns its name from the massive fiber bundles that cross its ventral surface, linking the cerebral cortex to the cerebellum. This region has two distinct parts: the basilar pons ventrally, dominated by pontine nuclei and transverse crossing fibers, and the pontine tegmentum dorsally, continuous with the medullary tegmentum and containing cranial nerve nuclei and ascending pathways. The basilar pons receives cortical projections via the cerebral peduncles, relays this information through the middle cerebellar peduncle—the largest of the three cerebellar connections—to the cerebellum for movement coordination.
Four cranial nerves have their nuclei in the pons. The trigeminal nerve (V), the largest cranial nerve, provides sensation to the face and powers the muscles of mastication. The abducens nerve (VI) turns the eye laterally—its long course makes it vulnerable to increased intracranial pressure. The facial nerve (VII) controls facial expression and carries taste from the anterior two-thirds of the tongue. The vestibulocochlear nerve (VIII) transmits hearing and balance information from the inner ear. Deep in the pontine tegmentum lie two small but mighty nuclei: the locus coeruleus, the brain's primary source of norepinephrine and crucial for arousal and attention, and the raphe nuclei, the main factory for serotonin.
The cerebellum attaches to the brainstem via three paired cerebellar peduncles—literally "little feet"—each carrying distinct information. The inferior peduncle brings input from the spinal cord (via spinocerebellar tracts) and medulla (via the inferior olive), providing proprioceptive and error-correction information. The middle peduncle, the largest, carries cortical input relayed through the pontine nuclei, allowing the cerebellum to know what movements the cortex intends. The superior peduncle carries cerebellar output to the red nucleus and thalamus, ultimately influencing motor cortex. This input-output organization makes the cerebellum a comparator: it receives information about intended movement and actual movement, calculates the error, and sends corrections. Damage to the cerebellum doesn't paralyze—it produces ataxia, the inability to coordinate smooth, accurate movement.
Reticular Formation and Ascending Arousal System
[VIEW IMAGES: Reticular formation and ascending arousal pathways]
Running through the core of the brainstem from medulla to midbrain, a diffuse network of neurons called the reticular formation earned its name from its net-like appearance—neurons with long, intermingling dendrites that create a reticulated web. This isn't a single structure but a collection of nuclei with diverse functions, including motor control, cardiovascular regulation, pain modulation, and most famously, the control of consciousness itself. The reticular formation is where ancient survival functions meet the modern brain—it decides whether you're awake enough to know you exist.
The ascending reticular activating system (ARAS) is the consciousness switch. Neurons in the upper brainstem reticular formation project to the cortex through two pathways: a dorsal pathway through the thalamus, particularly the intralaminar nuclei, which broadcast arousal signals to widespread cortical areas; and a ventral pathway through the hypothalamus and basal forebrain, which provides additional wake-promoting input. Stimulate the ARAS and an unconscious animal awakens; lesion it and consciousness disappears into coma. The ARAS explains why brainstem strokes can produce such devastating unconsciousness while leaving the cortex structurally intact—the cortex may be undamaged, but without the ARAS to wake it up, it remains silent.
Key components of the ascending arousal system include cholinergic neurons in the pedunculopontine (PPT) and laterodorsal tegmental (LDT) nuclei, which promote both waking and REM sleep; noradrenergic neurons in the locus coeruleus, which fire during waking and fall silent during sleep; serotonergic neurons in the raphe nuclei, which follow a similar pattern; and histaminergic neurons in the hypothalamic tuberomammillary nucleus, which are most active during waking. The orchestrated activity of these systems—rising and falling in coordinated patterns—generates the daily rhythm of waking and sleep, the moment-to-moment fluctuations of alertness, and the boundary between conscious experience and oblivion.
Midbrain: Colliculi, PAG, Substantia Nigra, Red Nucleus
[VIEW IMAGES: Midbrain structures]
The midbrain—the smallest brainstem segment—surrounds the narrow cerebral aqueduct and divides into three regions with distinct functions. The tectum (Latin for "roof") forms the dorsal midbrain, the tegmentum (Latin for "covering") forms the floor, and the paired cerebral peduncles constitute the ventral portion, carrying descending motor fibers from cortex to brainstem and spinal cord. Each region houses structures crucial for sensory processing, motor control, and the modulation of pain and behavior.
The tectum consists of four hillocks collectively called the corpora quadrigemina—two superior colliculi and two inferior colliculi. The superior colliculi receive direct retinal input and coordinate eye movements, particularly the rapid saccades that shift gaze from one target to another. They also generate orienting responses—the reflexive turn toward a sudden visual stimulus—that evolved long before the elaborate visual cortex that now dominates our visual processing. The inferior colliculi serve as obligatory relay stations in the auditory pathway, receiving input from lower auditory nuclei and projecting to the medial geniculate nucleus of the thalamus. Surrounding the cerebral aqueduct, the periaqueductal gray (PAG) is your body's built-in pain control center, containing endogenous opioid neurons that can suppress pain signals ascending from the spinal cord. The PAG also organizes defensive behaviors: stimulation of different regions produces freezing, flight, or fighting responses.
The tegmentum contains several crucial nuclei for motor control and motivation. The substantia nigra ("black substance," named for the dark neuromelanin pigment in its neurons) has two parts with opposite functions: the pars compacta contains dopaminergic neurons that project to the striatum and degenerate in Parkinson's disease, while the pars reticulata serves as a basal ganglia output nucleus. The red nucleus—pinkish due to its iron content—gives rise to the rubrospinal tract and participates in motor coordination, particularly of the upper limbs. The ventral tegmental area (VTA), adjacent to the substantia nigra, sends dopaminergic projections to the nucleus accumbens and prefrontal cortex—pathways crucial for reward, motivation, and the neurobiology of addiction. Two cranial nerve nuclei reside in the midbrain: the oculomotor nerve (III), controlling most eye movements and pupil constriction, and the trochlear nerve (IV), moving the superior oblique muscle to intort and depress the eye.
Ascending from the brainstem, we enter the diencephalon—the "between brain" that emerged from the embryonic prosencephalon. Here sit the thalamic nuclei that relay nearly every sensation to the cortex and the hypothalamic nuclei that maintain the body's internal environment. The diencephalon exemplifies the reciprocal principle: thalamus and cortex are locked in bidirectional loops, each influencing the other continuously.
Part VII: Diencephalic Nuclei
Thalamic Nuclei: Relay, Motor, and Association
[VIEW IMAGES: Thalamic nuclei and their cortical connections]
The thalamus—forming 80% of the diencephalon—is not a single nucleus but a collection of nuclei that serve as the grand central station of the brain. Nearly all sensory information (with the notable exception of olfaction) passes through specific thalamic nuclei before reaching the cortex. But the thalamus does far more than passively relay: it filters, gates, and synchronizes information, deciding what deserves cortical attention and what should be suppressed. Think of the thalamus as both a switchboard and a bouncer, routing calls while keeping out the riffraff.
The sensory relay nuclei demonstrate this organization most clearly. Visual information from the retina arrives at the lateral geniculate nucleus (LGN), which projects to primary visual cortex (V1). Auditory information reaches the medial geniculate nucleus (MGN), projecting to primary auditory cortex (A1). Somatosensory information from the body ascends to the ventral posterior nucleus—specifically the VPL for the body and VPM for the face—before reaching primary somatosensory cortex (S1). Each nucleus maintains the topographic organization of its sensory input, preserving spatial relationships as information is passed upward.
Motor-related nuclei receive input from cerebellum and basal ganglia and project to motor cortices. The ventral lateral nucleus (VL) receives cerebellar output via the superior cerebellar peduncle and projects to primary motor cortex, while the ventral anterior nucleus (VA) receives basal ganglia output and projects to premotor cortex. Association nuclei—including the massive pulvinar connecting with parietal and temporal association cortex for visual attention, the mediodorsal nucleus (MD) linking to prefrontal cortex for executive function, and the lateral posterior nucleus (LP) connecting to parietal cortex—integrate higher cognitive functions. The anterior nuclei participate in the Papez circuit for memory, connecting mammillary bodies to cingulate cortex. Wrapping around the thalamus like a shell, the reticular nucleus modulates all thalamic activity through inhibition, gating what information reaches the cortex and generating the sleep spindles that characterize light sleep. Critically, every thalamocortical connection is reciprocal—the cortex sends back as much as it receives, creating loops of bidirectional communication essential for perception and consciousness.
Hypothalamic Nuclei: Homeostatic Control
[VIEW IMAGES: Hypothalamic nuclei and their functions]
Weighing only about four grams—less than 1% of brain mass—the hypothalamus controls virtually everything that keeps you alive and reproducing. This almond-sized region below the thalamus serves as the body's thermostat, clock, hunger meter, thirst gauge, and hormone controller. Its numerous nuclei each specialize in specific homeostatic functions, working together to maintain the stable internal environment that complex life requires.
The suprachiasmatic nucleus (SCN), sitting just above the optic chiasm, is the master circadian pacemaker—receiving direct retinal input to entrain to the light-dark cycle and setting the body's 24-hour rhythms. The supraoptic nucleus (SON) and paraventricular nucleus (PVN) produce the hormones vasopressin (for water balance) and oxytocin (for bonding and lactation), sending axons directly to the posterior pituitary for release into the bloodstream. The PVN also regulates the stress response via the hypothalamic-pituitary-adrenal (HPA) axis. The lateral hypothalamus, the "feeding center," contains orexin neurons crucial for wakefulness—their destruction causes narcolepsy, while their silence permits normal sleep. Lesions here cause anorexia and lethargy. The ventromedial nucleus (VMH), the "satiety center," produces feelings of fullness; its destruction causes insatiable hyperphagia and obesity. The arcuate nucleus senses metabolic signals and produces releasing hormones that control the anterior pituitary, while the preoptic area regulates temperature and, through the ventrolateral preoptic nucleus (VLPO), promotes sleep.
The hypothalamus connects to the pituitary gland via the infundibulum, creating a crucial interface between nervous system and endocrine system. The posterior pituitary (neurohypophysis) is actually neural tissue—axon terminals of hypothalamic neurons that release oxytocin and vasopressin directly into the bloodstream. The anterior pituitary (adenohypophysis) is glandular tissue controlled by hypothalamic releasing and inhibiting hormones delivered via the hypophyseal portal system—a specialized vascular connection that carries hypothalamic signals to pituitary secretory cells. Through this two-part system, the hypothalamus ultimately controls the thyroid, adrenal cortex, gonads, and growth—making this tiny region the master regulator of the body's hormonal environment.
Epithalamus: Habenula and Pineal
The epithalamus, the "upper room" of the diencephalon, contains two small but significant structures. The habenular nuclei, receiving input from limbic structures and projecting to midbrain monoamine systems, participate in reward processing and aversive behavior—they're increasingly recognized as important nodes in the circuitry of depression and addiction. The pineal gland—the brain's only unpaired midline structure, which René Descartes mistakenly thought was the seat of the soul—produces melatonin in response to darkness. This hormone signals nighttime to the body, helping entrain circadian rhythms to the light-dark cycle. The pineal receives input indirectly from the SCN via the sympathetic nervous system—when the SCN's clock says "night" and light is absent, sympathetic activity decreases and melatonin secretion rises, inducing sleepiness and signaling to every organ that darkness has fallen.
Buried within each cerebral hemisphere, between the cortex above and the diencephalon below, lie the basal ganglia—deep nuclei that form loops with the cortex to select movements and shape habits. These structures provide yet another example of reciprocal organization: cortex projects to basal ganglia, which project through thalamus back to cortex, creating feedback circuits that modulate behavior at every moment.
Part VIII: Basal Ganglia and Deep Structures
Striatum and Motor Circuits
[VIEW IMAGES: Basal ganglia structures and connections]
Deep within each cerebral hemisphere lies a collection of interconnected nuclei that form loops with the cortex to control movement, motivation, and habit formation—the basal ganglia. These structures don't initiate movement the way motor cortex does; rather, they select which movements to allow and which to suppress, adjusting the gain on motor output based on context and reward history. The major input structure, the striatum, receives massive cortical projections and sets the tone for all downstream processing.
The striatum comprises three main components, each receiving distinct cortical inputs that determine their functional specialization. The caudate nucleus, a C-shaped structure whose head bulges into the lateral ventricle, receives primarily frontal and limbic cortical input and participates in cognitive and motivational functions. The putamen, lateral to the caudate, receives primarily sensorimotor cortical input and plays a central role in motor control. Though separated by the internal capsule, the caudate and putamen are actually continuous anteriorly, forming what's called the corpus striatum—the name "striatum" referring to the striped appearance created by fiber bundles passing through. The nucleus accumbens, the ventral striatum, receives input from limbic cortex and serves as the "reward center" central to motivation, pleasure, and the neurobiology of addiction.
Information flows from the striatum through output nuclei—the globus pallidus internal segment (GPi) and substantia nigra pars reticulata (SNr)—which send tonically active inhibitory (GABAergic) projections to the thalamus. Two parallel pathways modulate this output in opposite directions. The direct pathway runs from striatum to GPi/SNr: when striatal neurons fire, they inhibit the output nuclei, releasing the thalamus from inhibition and facilitating movement—a double-negative that becomes a positive. The indirect pathway interposes the globus pallidus external segment (GPe) and subthalamic nucleus between striatum and output nuclei, creating a circuit that increases output nucleus activity and suppresses movement. Dopamine from the substantia nigra pars compacta modulates these pathways in opposite directions: D1 receptors excite direct pathway neurons while D2 receptors inhibit indirect pathway neurons—both effects favor movement. The loss of dopamine neurons in Parkinson's disease tips this balance toward excessive inhibition, producing bradykinesia and rigidity; the loss of striatal neurons in Huntington's disease tips it toward insufficient inhibition, producing the involuntary movements called chorea.
Claustrum: The Mystery Structure
[VIEW IMAGES: Claustrum location and connections]
Sandwiched between the putamen medially and the insular cortex laterally lies the claustrum—a thin, irregular sheet of gray matter that remains one of neuroscience's most enigmatic structures. Despite its obscure location and small size, the claustrum may be the most densely interconnected structure in the brain, receiving from and projecting to nearly all cortical areas. Francis Crick, co-discoverer of DNA's structure, spent his final years studying the claustrum and proposed it might serve as the "conductor of consciousness," coordinating the activity of diverse brain regions into unified experience. This hypothesis remains unproven, but recent studies suggest the claustrum plays roles in attention, salience detection, and rapid task switching—functions that would indeed require integrating information across widely distributed cortical networks.
Internal Capsule and Corona Radiata
[VIEW IMAGES: Internal capsule and corona radiata]
Connecting the cerebral cortex to all subcortical structures and the spinal cord, the internal capsule is a thick band of white matter that passes between the basal ganglia nuclei. On its medial side lie the thalamus and caudate nucleus; on its lateral side sit the putamen and globus pallidus. This compact funnel concentrates fibers from the vast cortical surface into a narrow space, making it a vulnerable chokepoint where small lesions can produce devastating deficits.
The internal capsule has three parts, each carrying specific fiber types. The anterior limb, between the head of the caudate and the lentiform nucleus (putamen plus globus pallidus), carries frontopontine fibers and anterior thalamic radiations connecting thalamus to prefrontal cortex. The genu—the bend in the capsule—contains corticobulbar fibers descending to brainstem motor nuclei for face and tongue control. The posterior limb, between the thalamus and the lentiform nucleus, carries the crucial corticospinal fibers organized somatotopically—arm fibers anteriorly, leg fibers posteriorly—plus sensory radiations from the thalamus to the cortex. Above, the internal capsule fans out as the corona radiata, spreading to reach the entire cortical surface; below, it narrows into the cerebral peduncles as it descends through the midbrain.
The clinical importance of the internal capsule cannot be overstated. Because fibers from the entire motor cortex funnel through the posterior limb, a small stroke here—often from rupture of the thin-walled lenticulostriate arteries—produces dense contralateral hemiparesis affecting face, arm, and leg. The resulting "pure motor stroke" involves weakness without sensory loss, a pattern diagnostic of capsular involvement. The concentration of so many vital connections in such a small space represents one of the nervous system's few design flaws—a vulnerability inherited from the evolutionary expansion of the cortex faster than the white matter pathways could spread.
All roads lead to cortex. The cerebral cortex sits atop the neuraxis as the ultimate destination for ascending sensory pathways and the origin of descending motor commands. But the cortex is not a homogeneous sheet—it has its own intricate architecture, from the ancient allocortex of the limbic system to the six-layered neocortex that expanded explosively in primate evolution.
Part IX: Cortical Architecture
Nuclei vs Cortical Organization
[VIEW IMAGES: Nuclear vs laminar organization in the brain]
The brain organizes its neurons in two fundamentally different ways, each suited to different computational tasks. In nuclear organization, neurons cluster into discrete three-dimensional groups—nuclei—with similar functions, defined boundaries, characteristic cell types, and specific input-output connections. You can point to a thalamic nucleus, a brainstem nucleus, a basal ganglia nucleus, and say "that's the structure." Nuclei dominate subcortical organization and evolved first; they're the brain's ancestral architecture.
Cortical organization is radically different: neurons arrange themselves into layers—parallel sheets stacked like the pages of a book, each layer containing specific cell types with characteristic patterns of input and output connections. This laminar organization characterizes both the cerebral cortex and cerebellar cortex. The genius of layered organization is that it allows massive expansion of neural surface area through folding—sulci and gyri increase cortical volume without requiring proportional increases in skull size. Local circuit organization remains consistent from gyral crown to sulcal depth, but the folding multiplies processing capacity. The human cortex, unfolded, would cover about 2,500 square centimeters; folded, it fits within a skull that can pass through a birth canal.
Limbic Cortex: Allocortex and Transitional Zones
[VIEW IMAGES: Limbic system structures]
The limbic system—from the Latin limbus meaning "border" or "ring"—comprises a functional grouping of structures involved in emotion, memory, and motivated behavior. Unlike the neat anatomical divisions of brainstem or thalamus, the limbic system is defined by function rather than structure, including both cortical and subcortical components that form a ring around the upper brainstem and corpus callosum.
The cortical components include the cingulate cortex arching over the corpus callosum and involved in emotion regulation, error monitoring, and pain processing; the parahippocampal gyrus and entorhinal cortex participating in memory processing and spatial navigation; the orbitofrontal cortex evaluating reward and guiding decision-making; and the insular cortex processing interoception, emotion, and autonomic regulation. Subcortical components include the hippocampus—essential for forming new declarative memories, damage to which produces devastating anterograde amnesia; the amygdala—processing emotion, particularly fear, whose damage renders patients unable to recognize threatening faces or situations; the septal nuclei involved in reward and pleasure; and the mammillary bodies forming part of the Papez memory circuit.
Many limbic structures have allocortex—cortex with fewer than the six layers found in neocortex—or transitional cortex, reflecting their evolutionary age. The hippocampus has just three layers; the entorhinal cortex has an unusual structure with a cell-sparse lamina dissecans. These older cortical architectures predate the six-layered neocortex that dominates the human brain, serving functions—emotion, memory, basic motivation—that evolved before abstract reasoning and language. The limbic system reminds us that beneath our sophisticated cognition lies an ancient emotional brain that still drives much of our behavior.
Neocortex: Six-Layer Architecture
[VIEW IMAGES: Neocortical six-layer organization]
The neocortex—the "new cortex" that distinguishes mammals from other vertebrates—comprises approximately 80% of human brain mass. This vast neural expanse is extensively folded into gyri (ridges) and sulci (grooves), with particularly deep grooves called fissures. The folding isn't random: certain sulci are consistent landmarks present in every human brain—the central sulcus always separating motor from sensory cortex, the lateral (Sylvian) fissure always separating temporal from frontal and parietal lobes, the parieto-occipital sulcus marking the boundary between parietal and occipital cortex.
Despite regional variations, the neocortex maintains a six-layered architecture throughout, numbered I through VI from the pial surface to the white matter. Layer I, the molecular layer, contains few neurons but many dendrites and axons, serving as a zone where feedback connections from higher areas arrive. Layers II and III, the external granular and external pyramidal layers, contain small and medium pyramidal neurons that project to other cortical areas—the source of corticocortical connections that link different regions. Layer IV, the internal granular layer, contains spiny stellate cells and is the primary recipient of thalamic input—this layer is thick in sensory cortex (termed "granular cortex") where thalamic input dominates, and thin or absent in motor cortex (termed "agranular cortex") where output dominates. Layer V, the internal pyramidal layer, contains the largest pyramidal neurons—including the giant Betz cells of motor cortex—projecting to subcortical structures: striatum, brainstem, and spinal cord. Layer VI, the multiform layer with various cell types, projects back to the thalamus, completing the reciprocal loop between cortex and its relay station.
Cortical Columns: Functional Modules
[VIEW IMAGES: Cortical column organization]
Beyond its horizontal layers, the cortex is organized vertically into cortical columns—cylindrical units running perpendicular to the surface in which neurons at all layers process related information. Vernon Mountcastle discovered this principle in the 1950s when he found that neurons in a vertical column through somatosensory cortex all respond to the same sensory modality from the same body location. Insert an electrode perpendicular to the surface, and every neuron you encounter—from layer II through layer VI—responds to the same type of stimulus in the same place. Insert it at an angle, and you cross into columns with different response properties.
David Hubel and Torsten Wiesel extended this principle to visual cortex, discovering orientation columns (neurons preferring the same orientation of visual stimuli), ocular dominance columns (neurons preferring input from one eye or the other), and color blobs (clusters processing color information). Minicolumns—the smallest functional units at 40-50 micrometers wide containing 80-120 neurons—represent the basic processing module, while macrocolumns at 300-600 micrometers group minicolumns with similar properties into larger functional units. This columnar organization allows the cortex to repeat the same basic circuit across different areas while varying inputs and outputs—a modular design that evolution can modify by changing what a column receives and where it sends, without redesigning the basic computational unit.
Understanding cortical architecture sets the stage for our final topic: how do we map function onto this structure? From Brodmann's century-old cytoarchitectonic atlas to modern fMRI studies, neuroscience has progressively revealed which cortical regions do what—and how they connect to form the integrated networks that generate mind from matter.
Part X: Mapping and Connecting the Cortex
Brodmann Areas and Cytoarchitecture
[VIEW IMAGES: Brodmann area map]
In 1909, the German neurologist Korbinian Brodmann published a map that remains fundamental to neuroscience over a century later. By carefully examining the cellular architecture—the cytoarchitecture—of the cerebral cortex in histological sections, Brodmann divided it into 52 distinct Brodmann areas, each with characteristic cell types, densities, and layer patterns. Remarkably, many of these cytoarchitecturally defined regions correspond precisely to functionally distinct areas identified a century later with neuroimaging—a testament to the relationship between cellular structure and computational function.
Area 4, the primary motor cortex in the precentral gyrus, has a distinctive agranular structure with giant Betz cells in layer V projecting to the spinal cord. Area 6, immediately anterior, contains the premotor and supplementary motor areas involved in movement planning. Areas 1, 2, and 3 in the postcentral gyrus comprise primary somatosensory cortex, receiving thalamic input about touch and proprioception. Area 17, the primary visual cortex in the occipital pole, has a distinctive stripe of Gennari visible to the naked eye and elaborate layer IV specializations for processing visual input from the lateral geniculate nucleus. Areas 41 and 42 in the superior temporal gyrus contain primary auditory cortex. Areas 44 and 45 in the inferior frontal gyrus of the dominant hemisphere comprise Broca's area, essential for speech production—damage here produces non-fluent aphasia. Area 22 in the superior temporal gyrus includes Wernicke's area, where damage impairs speech comprehension. Areas 9, 10, and 46 in the prefrontal cortex underlie executive function, working memory, and personality—the regions Phineas Gage's tamping iron destroyed.
fMRI and Modern Functional Mapping
[VIEW IMAGES: fMRI activation maps]
Functional MRI (fMRI) has revolutionized our ability to map brain function in living humans. By measuring the BOLD (blood-oxygen-level-dependent) signal—changes in blood oxygenation that correlate with neural activity—fMRI reveals which brain regions activate during specific tasks. This technology has confirmed Brodmann's structural-functional correspondence while discovering new functional specializations invisible to cytoarchitecture.
fMRI has revealed the fusiform face area (FFA) in the fusiform gyrus, specialized for face processing—damage here produces prosopagnosia, the inability to recognize faces despite intact vision. The parahippocampal place area (PPA) responds selectively to scenes and buildings; the visual word form area (VWFA) activates specifically to written words. Perhaps most surprising was the discovery of the default mode network (DMN)—a set of regions including medial prefrontal cortex, posterior cingulate, and angular gyrus that become more active during rest than during task performance. This network, active during introspection, autobiographical memory, and self-referential thought, represents the brain's activity when it's not focused on the external world.
Primary, Secondary, and Association Cortex
The cortex is organized hierarchically, with information flowing from primary areas through secondary and association cortex to higher integrative regions, and back again. Primary cortex—V1 for vision, A1 for audition, S1 for somatic sensation, M1 for motor output—receives direct thalamic sensory input or provides the most direct motor output. Neurons in primary areas have simple receptive fields responding to basic features: edges and orientation in V1, specific frequencies in A1, touch location and modality in S1. Secondary cortex—V2, V3, and beyond for vision; secondary auditory and somatosensory areas—receives input from primary cortex and constructs more complex representations. Association cortex—parietal, temporal, and frontal areas beyond the primary and secondary zones—integrates across modalities, relates current information to memory and motivation, and formulates action plans.
Information flows both feedforward (up the hierarchy, from primary to association) and feedback (down the hierarchy, from association to primary). Perception requires both: bottom-up sensory information constrained by top-down predictions and expectations. This bidirectional flow explains why what you expect influences what you perceive, why context changes interpretation, and why perception is always an active construction rather than a passive reception of sensory data.
Corpus Callosum and Hemispheric Connections
[VIEW IMAGES: Corpus callosum and commissural connections]
The corpus callosum—containing approximately 200 million axons—is the brain's largest white matter structure, connecting homologous regions of the two cerebral hemispheres. This massive commissure allows the hemispheres to share information and coordinate activity, enabling unified perception and action despite the brain's fundamental bilateral organization. The corpus callosum has distinct regions connecting different cortical areas: the rostrum and genu connect the frontal lobes; the body connects motor, somatosensory, and parietal areas; and the splenium connects the occipital and temporal lobes.
Above the corpus callosum on the medial surface lies the cingulate cortex, a key limbic structure; behind the splenium lies the precuneus, a posterior midline region involved in self-referential processing and part of the default mode network. Despite largely symmetric structure, the hemispheres show functional lateralization: in most right-handers, the left hemisphere dominates for language while the right hemisphere specializes in spatial processing and face recognition. Studies of split-brain patients—whose corpus callosum was severed to treat epilepsy—revealed the independent processing capabilities of each hemisphere and demonstrated how the callosum normally integrates their separate contributions into unified experience. Beyond the corpus callosum, smaller commissures also connect the hemispheres: the anterior commissure linking temporal lobes and olfactory structures, the posterior commissure coordinating pupillary reflexes, and the hippocampal commissure connecting the two hippocampi.
Thought Questions for Discussion
The Redundancy Question: The circle of Willis provides redundancy in blood supply; bilateral structures provide redundancy in function. Yet many structures are unpaired (pineal, third ventricle) or show lateralization (language areas). What determines when evolution favors redundancy versus specialization? What are the tradeoffs?
The Segmentation Legacy: Spinal cord organization is clearly segmental—a legacy of our vertebrate ancestors. How much of brain organization reflects segmental origins? Consider that cranial nerves roughly follow segmental patterns, and brainstem nuclei are organized rostrocaudally. Has evolution fully escaped segmentation in building the brain?
The Feedback Paradox: We've emphasized reciprocal connections throughout—cortex to thalamus and back, cortex to basal ganglia and back. What computational advantages does this bidirectional organization provide? Could a purely feedforward system achieve what the brain achieves? Consider how artificial neural networks are increasingly incorporating recurrent connections.
Practice Questions:
• CSF flows from lateral ventricles through the interventricular foramina of _______ to the third ventricle, then through the cerebral aqueduct of _______ to the fourth ventricle.
• The five secondary brain vesicles from rostral to caudal are: _______, _______, _______, _______, and _______.
• The spinal cord ends at the _______ at vertebral level _______, with nerve roots below forming the _______.
• Dorsal column fibers synapse in the _______ and _______ nuclei of the medulla before crossing as the _______ lemniscus.
• The reticular activating system originates in the _______ and projects to cortex through the thalamus and _______.
• The substantia nigra pars compacta produces _______ and projects to the _______; its degeneration causes _______ disease.
• The direct pathway through basal ganglia involves striatum inhibiting _______, which disinhibits _______, facilitating movement.
• The six layers of neocortex are numbered I-VI from _______ to _______; layer _______ receives thalamic input while layer _______ projects to subcortical structures.
• Brodmann area 4 is _______ cortex, while areas 1, 2, and 3 comprise _______ cortex.
• The corpus callosum's genu connects _______ lobes while its splenium connects _______ lobes.
📝 View Answer Key for Chapter 5 Part 2
[PREVIEW: Sensory transduction and the construction of perception from sensation]