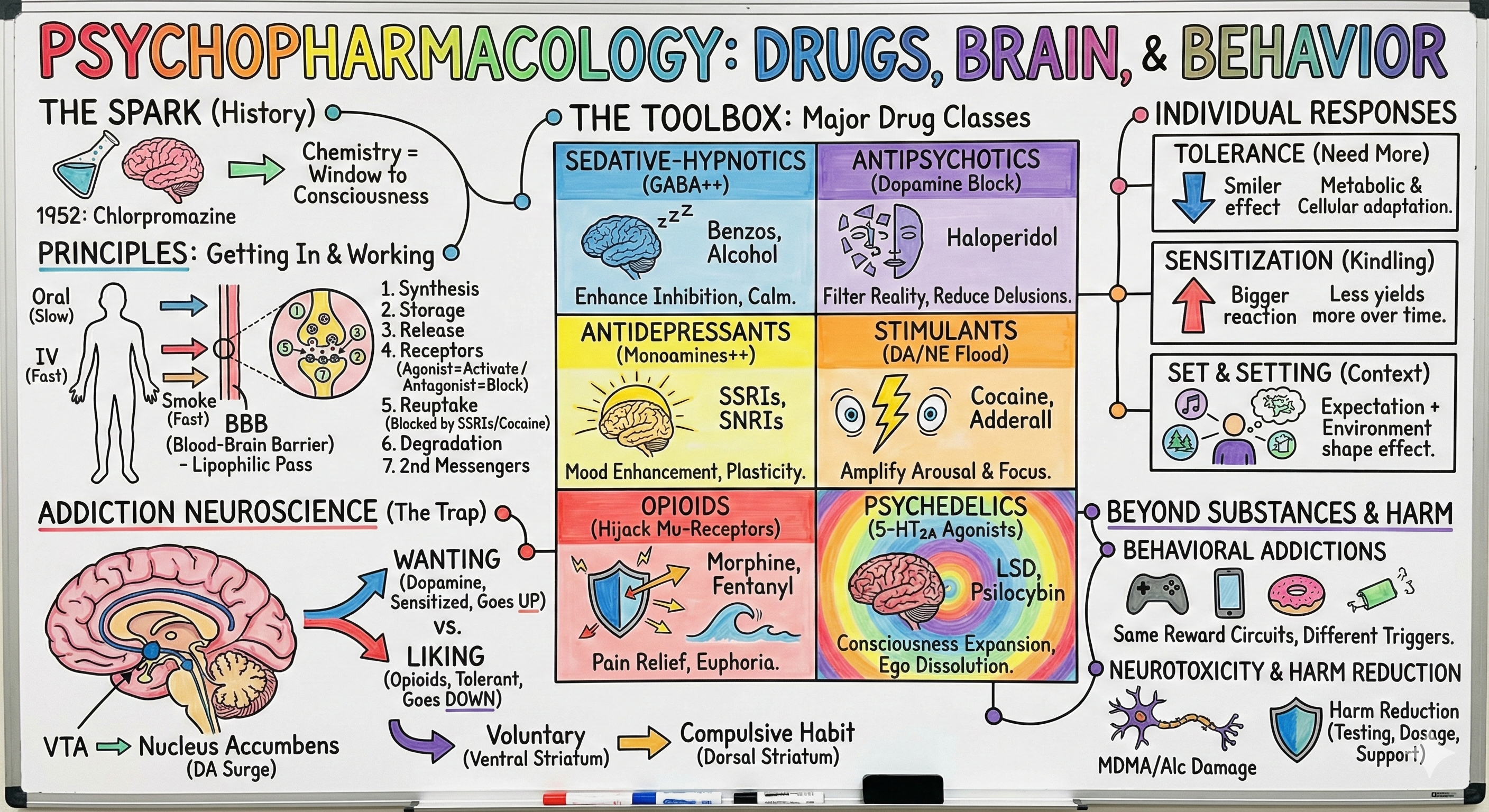
Psychopharmacology: Drugs, Brain, and Behavior
• Chlorpromazine and the birth of psychopharmacology
• Why drugs are essential tools for understanding brain function
• Chemistry as a window into consciousness
II. Principles of Psychopharmacology (15 min)
• Routes of administration and the blood-brain barrier
• Seven ways drugs modify synaptic transmission
• Agonists vs antagonists across neurotransmitter systems
• Drug metabolism and elimination
III. Major Classes of Psychoactive Drugs (18 min)
• Sedative-hypnotics: GABA enhancement and consciousness suppression
• Antipsychotics: Dopamine blockade and reality restoration
• Antidepressants: Monoamine hypothesis and beyond
• Stimulants: Dopamine and norepinephrine amplification
• Opioids: Endogenous pleasure systems hijacked
• Psychedelics: Serotonin 2A receptors and consciousness expansion
IV. Individual Responses: Tolerance, Sensitization, and Context (12 min)
• Metabolic, cellular, and learned tolerance
• Sensitization and the kindling effect
• Set and setting: How context shapes drug effects
• Genetic and sex differences in drug response
V. The Neuroscience of Addiction (15 min)
• Wanting vs liking: Incentive-sensitization theory
• Mesolimbic dopamine: The common pathway
• From voluntary control to compulsive habit
• Neuroplasticity in addiction: What Chapter 4 taught us
VI. Beyond Substances: Behavioral Addictions (8 min)
• Video games, gambling, and dopamine hits
• Sex, sugar, and social media: Natural rewards gone wrong
• Same circuits, different triggers
VII. Neurotoxicity and Harm Reduction (4 min)
• MDMA and serotonergic terminal damage
• Alcohol, thiamine deficiency, and Korsakoff's syndrome
• Glutamate excitotoxicity
Every thought you've ever had, every emotion you've felt, every decision you've made—all are chemical events in your brain. Psychoactive drugs are precise tools that allow us to manipulate these chemical systems and observe what happens to consciousness, behavior, and self. Today we'll explore how a few milligrams of molecules can transform depression into joy, psychosis into sanity, or normal consciousness into transcendent experience. You'll discover why the same drug can have opposite effects depending on dose and context, how addiction hijacks the learning systems we studied in Chapter 4, and why behavioral addictions—to video games, sugar, or social media—activate the same neural circuits as cocaine. From the accidental discovery of the first antipsychotic to MDMA-assisted psychotherapy, from caffeine's molecular mechanism to why tolerance develops, we'll trace how drugs reveal the chemical architecture of mind itself.
Opening: The Accident That Launched Modern Psychiatry
[VIEW IMAGES: Henri Laborit and the discovery of chlorpromazine in 1952]
In 1952, French surgeon Henri Laborit was searching for a drug to prevent surgical shock. He tried chlorpromazine, a compound synthesized by pharmaceutical chemists looking for better antihistamines. The drug didn't prevent shock, but Laborit noticed something extraordinary: his patients became calm and indifferent to their surroundings without losing consciousness. They could answer questions but seemed emotionally detached, describing a pleasant feeling of not caring. Laborit immediately recognized the psychiatric potential.
Within months, psychiatrists Jean Delay and Pierre Deniker administered chlorpromazine to patients with schizophrenia. The results were revolutionary. Patients who had been psychotic for years became lucid. Hallucinations and delusions diminished or disappeared. Asylums that had housed thousands of chronic patients began discharging them. Chlorpromazine became the first effective antipsychotic medication, launching the era of psychopharmacology and fundamentally changing how we understand and treat mental illness.
The discovery was serendipitous—nobody predicted that blocking a particular receptor would alleviate psychosis—but the implications were profound. If a chemical could eliminate delusions, then delusions must have a chemical basis. If molecules could restore sanity, then sanity itself was a chemical state. The mind-brain problem, which had been philosophical territory for millennia, suddenly became tractable through chemistry. Every major class of psychiatric medication was discovered through similar accidents over the next two decades: antidepressants while testing tuberculosis drugs, benzodiazepines while searching for new dyes, lithium while using it as a salt substitute.
Today, psychopharmacology isn't just about treating illness—it's one of our most powerful tools for understanding brain function. When we know that blocking dopamine D2 receptors reduces psychosis, we learn something fundamental about the role of dopamine in reality testing. When we discover that enhancing serotonin transmission alleviates depression, we gain insight into mood regulation. When we observe that NMDA receptor antagonists produce dissociative states, we understand more about how the brain constructs coherent experience. Drugs are like biochemical screwdrivers, allowing us to systematically disassemble the mechanisms of consciousness.
Principles of Psychopharmacology: Getting to the Brain
Routes of Administration: Barriers and Bioavailability
[VIEW IMAGES: Drug routes of administration showing different pathways to the brain]
For a drug to affect the brain, it must cross multiple barriers, each reducing bioavailability—the proportion of administered drug that reaches systemic circulation. Oral administration is convenient but faces the most obstacles. The drug must survive stomach acid, cross intestinal epithelium, pass through the liver (where first-pass metabolism destroys much of the dose), enter the bloodstream, and finally cross the blood-brain barrier. With each barrier, roughly 90% of the drug is lost. What started as a 100mg dose might deliver only 0.1mg to the brain.
Intravenous injection bypasses the digestive system and liver, delivering drug directly to the bloodstream. This dramatically increases bioavailability and produces rapid onset—within seconds, the drug reaches the brain. This explains why intravenous heroin is more addictive than oral morphine, even though they become the same molecule in the body. The rapid onset creates a more intense experience and stronger association between drug-taking behavior and reward. Smoking and intranasal administration also provide rapid onset by crossing lung or nasal membranes directly into blood vessels.
Transdermal patches provide slow, steady delivery by diffusing through skin. This maintains stable blood levels, avoiding the peaks and troughs of repeated doses. Nicotine patches reduce craving by maintaining baseline receptor occupancy without the rapid spikes that create addiction. Intramuscular injection provides intermediate kinetics—faster than oral but slower than intravenous. In emergency psychiatry, intramuscular antipsychotics rapidly sedate agitated patients who can't take oral medication.
The most direct route is intracerebral injection—directly into brain tissue—used only in research. But even this faces the challenge that neurons aren't uniformly distributed. Injecting into cortex might affect completely different circuits than injection into hippocampus. The ideal drug delivery would target specific cell types in specific brain regions—a goal that optogenetics and chemogenetics are beginning to achieve.
The Blood-Brain Barrier: Guardian and Obstacle
[VIEW IMAGES: Blood-brain barrier structure showing tight junctions and astrocyte end feet]
Recall from Chapter 5 that the blood-brain barrier (BBB) protects the brain from toxins, pathogens, and fluctuations in blood chemistry. Endothelial cells lining brain capillaries are joined by tight junctions that prevent passive diffusion of most molecules. Astrocyte end feet cover about 80% of capillary surfaces, further regulating what enters and exits. Only small, lipophilic (fat-soluble), uncharged molecules like oxygen and carbon dioxide freely cross. Larger or charged molecules require active transport.
This protection creates a pharmaceutical problem: an estimated 98% of potential brain drugs cannot cross the BBB. The barrier is why L-DOPA, rather than dopamine itself, treats Parkinson's disease—dopamine is too polar to cross, but L-DOPA uses an amino acid transporter, then converts to dopamine inside the brain. It's why heroin is more potent than morphine—the acetyl groups make heroin more lipophilic, increasing BBB penetration. Once across, esterases remove the acetyl groups, yielding morphine.
Some brain regions lack the BBB. The area postrema in the medulla senses toxins in blood and triggers vomiting—you want this region exposed so you can expel poisons. The pineal gland needs access to hormones that regulate circadian rhythms. The median eminence releases hypothalamic hormones into the pituitary. These BBB-free regions are called circumventricular organs, and their exposure creates vulnerabilities—infections and toxins can enter through these portals.
Drug Metabolism and Elimination
The body treats drugs as foreign invaders and works to eliminate them. The liver is the primary metabolic site, using cytochrome P450 enzymes to break down drugs into more water-soluble metabolites that kidneys can excrete. Some drugs are metabolized to inactive forms (most benzodiazepines), while others produce active metabolites (codeine metabolizes to morphine, the actual analgesic). Occasionally, metabolism produces toxic metabolites—acetaminophen overdose generates a hepatotoxic metabolite that destroys liver cells.
The half-life is the time required for plasma concentration to decrease by half. Short half-life drugs (cocaine: 1 hour) require frequent dosing and produce rapid withdrawal, increasing addiction liability. Long half-life drugs (fluoxetine: 4-6 days) maintain steady levels with once-daily dosing and produce gentler withdrawal. Methadone's long half-life (24-36 hours) makes it useful for treating heroin addiction—it prevents withdrawal without producing the rapid high that maintains addiction.
Individual differences in P450 enzymes profoundly affect drug response. Poor metabolizers have genetic variants that reduce enzyme activity, causing drugs to accumulate to toxic levels at standard doses. Ultrarapid metabolizers break down drugs so quickly that therapeutic levels are never achieved. About 10% of Caucasians are poor metabolizers of drugs processed by CYP2D6, while 30% of North Africans are ultrarapid metabolizers. Pharmacogenomics—tailoring drug choice and dose to genetic profile—is beginning to address this variability.
How Drugs Modify Synaptic Transmission
Seven Synaptic Targets
[VIEW IMAGES: Seven ways drugs modify synaptic transmission]
Recall from Chapters 2 and 3 our detailed exploration of chemical transmission—action potentials trigger calcium influx, vesicles fuse, neurotransmitter diffuses across the gap, receptors activate, signals terminate through reuptake or degradation. Drugs can target any step in this cascade. Let's examine each intervention point:
1. Synthesis: Drugs can block neurotransmitter production. Alpha-methyl-para-tyrosine blocks tyrosine hydroxylase, preventing dopamine and norepinephrine synthesis. Within hours, these neurotransmitter stores deplete, producing sedation and depression. Conversely, L-DOPA increases dopamine synthesis by providing extra precursor. This demonstrates that neurotransmitter levels can limit transmission—more precursor means more transmitter means stronger signals.
2. Storage: Reserpine destroys vesicular storage, causing neurotransmitter to leak into cytoplasm where it's degraded by monoamine oxidase. Dopamine, norepinephrine, and serotonin stores deplete over days, producing profound depression. Reserpine was once used to treat hypertension but abandoned due to severe depressive side effects—side effects that helped establish the monoamine hypothesis of depression.
3. Release: Amphetamine reverses dopamine and norepinephrine transporters, causing massive neurotransmitter release. Instead of reuptake removing transmitter from the synapse, the transporter pumps it out. This floods synapses with neurotransmitter, producing euphoria, energy, and confidence. Botulinum toxin (Botox) cleaves SNARE proteins, preventing acetylcholine release at neuromuscular junctions. The result is paralysis—muscles can't contract without acetylcholine.
4. Receptor binding: Most drugs act here. Agonists activate receptors, mimicking the natural neurotransmitter. Morphine binds mu-opioid receptors, producing analgesia and euphoria. Nicotine binds nicotinic acetylcholine receptors, producing arousal and reward. Antagonists block receptors without activating them. Naloxone blocks opioid receptors, instantly reversing heroin overdose. Haloperidol blocks dopamine D2 receptors, reducing psychotic symptoms.
The key insight: agonists and antagonists can target the same receptor with opposite effects. Benzodiazepines are GABA-A receptor positive allosteric modulators—they don't activate the receptor directly but enhance GABA's effect when it binds. This produces the anxiolytic (anxiety-reducing) and sedative effects. Flumazenil, a benzodiazepine antagonist, blocks this enhancement, reversing benzodiazepine overdose.
5. Reuptake inhibition: If you block the transporter that removes neurotransmitter from the synapse, the neurotransmitter lingers, producing prolonged receptor activation. SSRIs (selective serotonin reuptake inhibitors) block the serotonin transporter, increasing synaptic serotonin and alleviating depression. Cocaine blocks dopamine, norepinephrine, and serotonin transporters, producing euphoria and energy. The same mechanism—reuptake inhibition—can treat illness or cause addiction depending on which transmitter, which circuits, and which kinetics.
6. Enzymatic degradation: Blocking the enzymes that break down neurotransmitters prolongs their action. Acetylcholinesterase inhibitors prevent acetylcholine breakdown, used in myasthenia gravis (to strengthen muscle contraction) and Alzheimer's disease (to enhance cholinergic transmission in memory circuits). Nerve agents like sarin are irreversible acetylcholinesterase inhibitors—they cause fatal overstimulation of muscles including the diaphragm.
7. Second messenger systems: Some drugs bypass the receptor and directly affect intracellular signaling. Lithium, used in bipolar disorder, affects inositol metabolism and GSK-3 beta activity, modulating multiple signaling cascades. The effects are complex and not fully understood, but they stabilize the dramatic mood swings of bipolar disorder.
Major Classes of Psychoactive Drugs
Sedative-Hypnotics: Enhancing Inhibition
[VIEW IMAGES: GABA receptor showing benzodiazepine binding sites]
Sedative-hypnotics reduce neural excitability, producing calming, sleep-inducing effects. The primary target is the GABA-A receptor, the main inhibitory receptor in the brain. Benzodiazepines (diazepam, alprazolam, lorazepam) bind to specific sites on GABA-A receptors, increasing the frequency of chloride channel opening when GABA binds. More chloride enters, more hyperpolarization occurs, neurons become less likely to fire. The result: reduced anxiety, muscle relaxation, sedation, amnesia.
Barbiturates also target GABA-A receptors but at a different site, increasing the duration of channel opening. They're more dangerous than benzodiazepines because at high doses they can directly activate receptors without GABA, potentially causing fatal respiratory depression. Barbiturates were once widely used as sleeping pills but have been largely replaced by safer alternatives. Alcohol (ethanol) enhances GABA transmission through multiple mechanisms while also affecting glutamate, dopamine, serotonin, and opioid systems—its widespread effects explain why alcohol intoxication is so complex.
The therapeutic index—the ratio between therapeutic and toxic doses—varies enormously. Benzodiazepines have a high therapeutic index; overdose rarely causes death unless combined with other depressants. Barbiturates and alcohol have low therapeutic indices; the difference between a sleep-inducing dose and a fatal dose is dangerously small. This is why mixing alcohol with benzodiazepines or opioids is so dangerous—the depressant effects summate, causing respiratory arrest.
Antipsychotics: Filtering Reality
Antipsychotic medications treat schizophrenia and other psychotic disorders characterized by hallucinations, delusions, and disorganized thinking. All effective antipsychotics share one property: they block dopamine D2 receptors. This discovery led to the dopamine hypothesis of schizophrenia—the proposal that psychosis involves excessive dopamine transmission in mesolimbic pathways. The hypothesis is oversimplified (schizophrenia involves many neurotransmitter systems), but dopamine clearly plays a central role.
[VIEW IMAGES: Dopamine pathways affected by antipsychotic medications]
First-generation (typical) antipsychotics like haloperidol and chlorpromazine strongly block D2 receptors. They reduce positive symptoms (hallucinations, delusions) but cause serious motor side effects because they also block D2 receptors in the nigrostriatal pathway controlling movement. Prolonged use can cause tardive dyskinesia—permanent involuntary movements. Second-generation (atypical) antipsychotics like clozapine and risperidone also block serotonin 5-HT2A receptors, reducing motor side effects while improving negative symptoms (flat affect, social withdrawal, cognitive deficits).
The effectiveness of antipsychotics reveals something profound about delusions and hallucinations: they're not just false beliefs or perceptions but arise from disrupted neural signaling. Block the right receptor, and reality testing restores. This doesn't mean schizophrenia is "just" a chemical imbalance—the causes involve genetics, development, stress, and social factors—but it does mean that chemistry mediates the expression of symptoms.
Antidepressants: Monoamine Enhancement and Beyond
Antidepressants treat major depression, anxiety disorders, OCD, and PTSD. The monoamine hypothesis proposes that depression involves deficient transmission of serotonin, norepinephrine, or dopamine. Evidence includes: reserpine (which depletes monoamines) causes depression, antidepressants increase monoamine transmission, and people with depression show altered monoamine metabolism. However, the hypothesis is incomplete—antidepressants increase monoamines within hours, but clinical improvement takes weeks, suggesting downstream neuroplastic changes are critical.
[VIEW IMAGES: Antidepressant mechanisms and monoamine systems]
SSRIs (fluoxetine, sertraline, escitalopram) selectively block serotonin reuptake. They're first-line treatments because of favorable side effect profiles. SNRIs (venlafaxine, duloxetine) block both serotonin and norepinephrine reuptake. NDRIs (bupropion) block norepinephrine and dopamine reuptake, useful for patients who don't respond to SSRIs. MAOIs (monoamine oxidase inhibitors) block the enzyme that degrades all monoamines. They're highly effective but rarely used due to dangerous interactions with certain foods containing tyramine.
Newer theories propose that antidepressants work by promoting neuroplasticity—the processes we studied in Chapter 4. Chronic stress reduces hippocampal neurogenesis, dendritic branching, and BDNF (brain-derived neurotrophic factor) levels. Antidepressants restore neurogenesis and increase BDNF, allowing depressed circuits to remodel. This explains the delayed therapeutic effect—synaptic changes and neuroplasticity take weeks to manifest. It also suggests non-drug treatments that promote plasticity (exercise, psychotherapy, sleep) should augment antidepressant effects.
The rapid antidepressant effects of ketamine and esketamine (NMDA receptor antagonists) have challenged the monoamine hypothesis. These drugs produce improvement within hours, long before neuroplastic changes could occur. They work through different mechanisms—enhancing AMPA receptor transmission and triggering rapid synaptic protein synthesis. This has sparked renewed interest in glutamate-based theories of depression and opened new treatment avenues.
Stimulants: Amplifying Arousal Systems
Stimulants increase dopamine and norepinephrine transmission, producing alertness, energy, euphoria, and improved focus. Amphetamine and methamphetamine cause neurotransmitter release and block reuptake, flooding synapses. Methylphenidate (Ritalin) and cocaine block reuptake without causing release. The effects are similar but kinetics differ—cocaine's rapid onset and short duration (smoked: peak in seconds, gone in minutes) make it highly addictive. Methylphenidate's slower onset and longer duration (oral: peak in 1-2 hours, lasting 3-4 hours) provide therapeutic benefits with lower addiction risk.
[VIEW IMAGES: Stimulant mechanisms showing dopamine release and reuptake blockade]
The paradox of ADHD treatment is that stimulants calm hyperactive children. The explanation relates to cortical control. ADHD involves underactive prefrontal cortex, reducing inhibitory control over impulsive behaviors. Stimulants activate prefrontal dopamine and norepinephrine receptors, strengthening top-down control. This allows better regulation of attention and behavior. The same drug can produce opposite behavioral effects depending on baseline brain state—a recurring theme in psychopharmacology.
Opioids: Hijacking Endogenous Pleasure
Your brain produces its own opioids—endorphins, enkephalins, and dynorphins—that activate opioid receptors (mu, delta, kappa). These endogenous opioids mediate pain relief, reward, and social bonding. Exogenous opioids like morphine, heroin, oxycodone, and fentanyl hijack this system. They bind mu-opioid receptors with high affinity, producing profound analgesia, euphoria, and respiratory depression (the cause of fatal overdose).
The locus coeruleus in the brainstem contains noradrenergic neurons that express dense mu-opioid receptors. Opioids inhibit these neurons, reducing norepinephrine release and producing the characteristic opioid relaxation. But chronic opioid use causes compensatory upregulation—more receptors, less endogenous opioid production. When the drug is withdrawn, the locus coeruleus hyperactivates, causing the physical withdrawal symptoms: muscle aches, sweating, nausea, anxiety. The psychological craving involves different circuits—mesolimbic dopamine pathways we'll examine in the addiction section.
[VIEW IMAGES: Opioid receptor distribution and endogenous opioid systems]
The opioid crisis—over 100,000 overdose deaths annually in the US—stems partly from fentanyl, 50-100 times more potent than morphine. A tiny amount produces respiratory arrest. Naloxone, an opioid antagonist, can reverse overdose by competitively blocking receptors, but requires prompt administration. The crisis illustrates how understanding receptor pharmacology translates directly to public health interventions.
Psychedelics: Consciousness Expansion Through Serotonin
[VIEW IMAGES: Psychedelic mechanisms showing 5-HT2A receptor activation and DMN disruption]
Psychedelics—LSD, psilocybin, mescaline, DMT—produce profound alterations in perception, thought, and self-awareness. All are 5-HT2A receptor agonists, primarily affecting cortical layer V pyramidal neurons. This causes increased neural entropy—more unpredictable, less constrained neural activity. Brain networks that normally communicate separately begin cross-talking, producing synesthesia (seeing sounds, hearing colors) and novel insights.
Recall from Chapter 12 that the default mode network (DMN) maintains self-referential processing and ego boundaries. Psychedelics suppress DMN activity, correlating with ego dissolution—the sense that the boundary between self and world has dissolved. Many users report this as profoundly meaningful, sometimes therapeutic. Recent studies show psilocybin-assisted therapy produces rapid, sustained improvements in treatment-resistant depression and anxiety, potentially by disrupting pathological thought patterns (like the rumination we learned about in depression) and allowing therapeutic reorganization.
MDMA (ecstasy) combines serotonin release with some psychedelic properties, producing empathy and emotional openness. It's being studied for PTSD treatment in combination with psychotherapy. The drug reduces fear and defensiveness, allowing patients to process traumatic memories without overwhelming anxiety. If approved, MDMA-assisted psychotherapy would represent a new treatment paradigm combining pharmacology with psychotherapy.
Individual Differences: Why the Same Drug Affects People Differently
Tolerance: When More Is Needed
Tolerance means that repeated drug administration produces progressively smaller effects—more drug is needed to achieve the same response. Three distinct mechanisms create tolerance. Metabolic tolerance involves increased liver enzyme production. The liver recognizes the drug as foreign and ramps up P450 enzyme synthesis. More enzyme means faster breakdown, requiring higher doses to maintain blood levels. Chronic alcohol drinkers develop metabolic tolerance, metabolizing alcohol much faster than naive users.
Cellular tolerance involves neurons adapting to minimize the drug's effects. If a drug chronically increases neurotransmitter levels, postsynaptic neurons downregulate receptors—fewer receptors mean less response to the same transmitter levels. If a drug blocks receptors, neurons upregulate receptor density and increase transmitter release, partially compensating for the blockade. These homeostatic mechanisms maintain neural function despite chronic drug presence, but create dependence—removing the drug now uncovers the adaptations.
[VIEW IMAGES: Mechanisms of tolerance showing receptor regulation]
Learned tolerance involves behavioral compensation. People learn to minimize intoxication's impairment through practice. Studies show that subjects given alcohol while performing tasks show initial impairment, but performance improves over days even with constant blood alcohol levels. They're not metabolizing faster or showing cellular tolerance—they're learning strategies to compensate for impairment. This explains why chronic drinkers can function at blood alcohol levels that would incapacitate naive users.
Sensitization: When Less Produces More
Sensitization is tolerance's opposite—repeated administration produces progressively larger effects. Behavioral sensitization occurs with many drugs of abuse, particularly stimulants. Rats given amphetamine every 3-4 days show increasing locomotor activation with each dose. The same dose that barely increased activity initially produces frantic running after several exposures. This reflects lasting changes in dopamine circuits—increased release, receptor sensitivity, or neural excitability.
Sensitization has dark implications for addiction. As sensitization develops, drug-associated cues trigger increasing craving and compulsive drug-seeking. The same environmental cues that initially produced mild craving eventually trigger overwhelming urges. This is kindling—progressive amplification of response with periodic exposure. It's the opposite of habituation (which we learn about in Chapter 10) where repeated stimuli produce decreasing responses. Whether tolerance or sensitization develops depends on dosing schedule (continuous vs periodic), drug class, and which neural systems are engaged.
Set and Setting: Context Is Everything
The same drug can produce vastly different subjective experiences depending on psychological set (expectations, mood, personality) and setting (physical and social environment). Indigenous peoples use ayahuasca (containing DMT) in ceremonial contexts with experienced guides, producing spiritual insights. The same drug taken recreationally in an unfamiliar environment can produce terrifying psychotic-like experiences. The molecules are identical; the context shapes everything.
[VIEW IMAGES: Set and setting influence on drug effects]
Placebo effects demonstrate how expectation modulates drug response. If you believe a drug will relieve pain, it often does, even if it's inert. Brain imaging shows placebo analgesia activates the same opioid circuits as real analgesics—expectation triggers endogenous opioid release. Conversely, nocebo effects occur when negative expectations worsen outcomes. If you expect a drug to cause nausea, it often does, even if it's inert.
The phenomenon of state-dependent learning shows that information learned in one drug state is better recalled in the same state. If you study while drinking coffee, you'll recall better during a caffeinated test than an uncaffeinated one. The drug state becomes part of the memory context. This has implications for treating addiction—interventions delivered during intoxication may not transfer to sober states.
Sex Differences and Genetic Variability
Women are typically more sensitive to drugs than men due to smaller body mass, higher body fat percentage (affecting lipophilic drug distribution), hormonal fluctuations affecting liver enzymes, and different receptor densities. Women progress from first use to addiction faster than men for cocaine, amphetamine, opioids, and alcohol—a phenomenon called "telescoping." Yet women are underrepresented in drug studies, potentially leading to suboptimal dosing and treatment.
Genetic variation in receptor genes, transporters, and metabolic enzymes creates enormous individual variability. Some people lack functional ALDH2 (aldehyde dehydrogenase), the enzyme that metabolizes alcohol's toxic metabolite acetaldehyde. Alcohol makes them feel terrible—flushing, nausea, rapid heartbeat—providing natural protection against alcoholism. Others have variants in mu-opioid receptor genes that alter opioid efficacy and addiction risk. Personalized medicine aims to match drugs to genetic profiles, but we're still early in understanding these complex gene-drug interactions.
The Neuroscience of Addiction: From Pleasure to Compulsion
Wanting vs Liking: The Incentive-Sensitization Theory
[VIEW IMAGES: Incentive-sensitization theory showing dissociation of wanting and liking]
Traditional views held that addiction results from seeking the drug's pleasurable effects and avoiding withdrawal. But this can't explain why addicts often report little pleasure from drugs yet continue using compulsively, or why former addicts relapse months after withdrawal symptoms have resolved. Terry Robinson and Kent Berridge's incentive-sensitization theory proposes that addiction involves dissociation between wanting and liking.
Liking is the pleasure derived from drugs, mediated primarily by opioid neuron activity in the nucleus accumbens shell and ventral pallidum. With repeated use, tolerance develops—the same dose produces less pleasure. This hedonic tolerance is why experienced users often report drugs aren't as good as they remember. Wanting (craving) is the motivation to obtain drugs, mediated by mesolimbic dopamine pathways from the ventral tegmental area (VTA) to nucleus accumbens. With repeated use, sensitization develops—cue-triggered dopamine release intensifies, producing overwhelming craving.
The tragedy of addiction is that wanting and liking go in opposite directions. You want the drug more and more but like it less and less. The gulf between motivation and reward expands until you're compulsively seeking something that no longer provides satisfaction. This explains why addicts continue using despite devastating consequences—the behavior is driven by sensitized wanting, not rational pursuit of pleasure.
The Mesolimbic Dopamine System: The Common Pathway
[VIEW IMAGES: Mesolimbic dopamine pathway showing addiction circuitry]
All addictive drugs—despite diverse chemical structures and receptor targets—share one property: they increase dopamine in the nucleus accumbens. Stimulants do this directly. Opioids disinhibit VTA dopamine neurons by inhibiting GABAergic interneurons. Nicotine activates nicotinic receptors on VTA neurons. Alcohol enhances dopamine release through multiple mechanisms. The nucleus accumbens acts as an interface between motivation and action, receiving input from amygdala (emotional significance), hippocampus (context and memory), and prefrontal cortex (goals and plans), then projecting to motor systems to generate behavior.
Dopamine doesn't signal pleasure per se—it signals reward prediction error, as we learned in the learning and memory chapter. When a reward is better than expected, dopamine neurons fire, strengthening connections that led to the reward. This is exactly what we learned about in Chapter 4—dopamine enables synaptic plasticity through its modulation of LTP and LTD. Drugs produce massive dopamine release—far exceeding natural rewards like food or sex—hijacking this learning system. The brain essentially learns that drug-taking is the most important behavior possible.
Drug-associated cues—the sight of a bar, the smell of marijuana, the people you used with—become powerfully associated with dopamine release through classical conditioning. Eventually, these cues trigger dopamine and craving without the drug being present. This cue-reactivity explains why former addicts relapse when exposed to drug-associated contexts. The conditioned responses are incredibly persistent; in rodents, they can last months without extinction.
From Voluntary Control to Compulsive Habit
The progression from recreational use to addiction involves a shift from ventral to dorsal striatum control. Initial drug use is voluntary and goal-directed, mediated by the ventral striatum (nucleus accumbens) and its connections with prefrontal cortex. You choose to use drugs for their effects. With repeated use, control shifts to the dorsal striatum—particularly the dorsolateral region involved in habit formation.
[VIEW IMAGES: Brain changes in addiction showing striatal transitions]
Habits are stimulus-response associations that occur automatically without conscious deliberation—like driving home while thinking about something else. When drug use becomes habitual, it's triggered by cues and contexts without conscious decision-making. This explains why addicts use despite consciously wanting to quit—the behavior has become automatized, controlled by dorsal striatal circuits that operate outside conscious awareness. Recovery requires reestablishing prefrontal cortex control over behavior, a slow process involving the neuroplasticity mechanisms we studied in Chapter 4.
Brain imaging studies of addicts show reduced prefrontal cortex activity and reduced gray matter volume in regions critical for inhibitory control, decision-making, and self-regulation. Some of these changes predate drug use (genetic vulnerability), while others result from chronic intoxication. Either way, the impaired prefrontal function creates a vicious cycle: reduced executive control makes it harder to resist cravings, leading to more use, causing more prefrontal damage.
Beyond Substances: Behavioral Addictions
When Natural Rewards Become Pathological
[VIEW IMAGES: Brain activity in behavioral addictions showing similar patterns to substance addiction]
If addiction involves dopamine signaling and neuroplasticity in reward circuits, then potentially any rewarding behavior could become addictive. Gambling disorder is now classified as an addiction because it shows the same features: tolerance (needing to bet more), withdrawal (irritability when unable to gamble), loss of control, continuation despite negative consequences, and similar patterns of brain activation. Pathological gamblers show hyperactivity in ventral striatum when viewing gambling cues and hypoactivity in prefrontal cortex during decision-making—the same pattern seen in substance addiction.
Video game addiction affects about 1-10% of gamers depending on criteria. Games are explicitly designed to provide variable ratio reinforcement schedules—the most addictive reinforcement pattern, as we learned in behavioral psychology. Random loot drops, level-ups, and achievement notifications all trigger dopamine release. Multiplayer games add social reinforcement. Brain imaging shows that gaming activates the same mesolimbic pathways as drugs, though typically with smaller magnitude. For vulnerable individuals—those with poor prefrontal control, high impulsivity, or social anxiety—gaming can escalate from hobby to compulsion.
Social media and smartphone addiction exploit the same reward circuits. Each notification, like, or message is a small dopamine hit. The variable schedule (you never know when the next reward arrives) maintains checking behavior. Features like Snapchat streaks or Instagram likes are deliberately engineered to be addictive. The average person checks their phone 96 times daily—once every 10 minutes during waking hours. This isn't accidental; it's the result of companies employing psychologists and neuroscientists to maximize engagement using principles of addiction.
Sugar: The Socially Acceptable Drug
[VIEW IMAGES: Sugar effects on brain reward systems]
Sugar activates opioid and dopamine systems in patterns remarkably similar to drugs of abuse. Rats given intermittent sugar access show bingeing, withdrawal, and craving behaviors. When naloxone (opioid antagonist) is administered, they show withdrawal symptoms—anxiety, teeth chattering, tremor. Cross-sensitization occurs: sugar-bingeing rats show enhanced responses to amphetamine, and amphetamine-sensitized rats consume more sugar. This suggests common neural mechanisms.
The modern food environment—with hyperpalatable foods engineered for maximum reward value—may constitute a drug-like challenge to our reward systems. Our ancestors encountered sweet foods rarely (honey, ripe fruit), so our brains evolved to find them highly rewarding. Modern processed foods deliver sugar concentrations never encountered in evolutionary history. The result: obesity rates have tripled since 1975, now affecting 40% of adults. While obesity is complex and multifactorial, the addictive properties of palatable foods certainly contribute.
Sex, Exercise, and Other Natural Highs
Sexual activity releases dopamine, opioids, and oxytocin—creating powerful reinforcement. For most people, this is adaptive. But some develop compulsive sexual behavior that impairs functioning—spending hours daily viewing pornography, engaging in risky sexual encounters despite negative consequences, or being unable to maintain relationships. Brain imaging shows hyperactivity in reward circuits when viewing sexual stimuli, similar to drug cues in addicts.
Exercise produces "runner's high" through endocannabinoid and endorphin release. For most, this motivates healthy behavior. But some develop exercise addiction—compulsive exercising despite injury, neglecting responsibilities, and experiencing withdrawal (anxiety, irritability) when unable to exercise. The same neuroplasticity that makes exercise beneficial for depression can, in extreme cases, create dependence.
The key insight: the addiction circuits evolved to reinforce behaviors essential for survival and reproduction—eating, sex, social bonding. Drugs hijack these circuits by producing supraphysiological activation. But modern environments present novel stimuli (processed foods, internet pornography, video games) that can also overstimulate these systems. The circuits don't distinguish between adaptive and maladaptive activation; they just learn to want whatever produces the strongest dopamine signal.
Neurotoxicity and Treatment
Do Drugs Damage the Brain?
[VIEW IMAGES: MDMA neurotoxicity showing serotonin terminal damage]
Many substances can be neurotoxic, but determining whether recreational drugs cause permanent damage is challenging. MDMA (ecstasy) provides the clearest evidence. Doses approximating human recreational use cause degeneration of fine serotonergic terminals in rodents and primates. PET imaging shows reduced serotonin transporter density in MDMA users, suggesting terminal loss. The damage may be permanent in some brain regions, though partial recovery occurs with abstinence.
Alcohol doesn't directly kill neurons, but chronic heavy use causes brain damage indirectly. Alcoholics often have poor nutrition and particularly lack thiamine (vitamin B1), essential for cellular metabolism. Wernicke-Korsakoff syndrome results from thiamine deficiency, causing lesions in thalamus and mammillary bodies. Patients develop profound amnesia, confabulation, and cognitive deficits. The syndrome is preventable with thiamine supplementation but often irreversible once damage occurs.
Methamphetamine causes dopamine terminal damage at high doses, likely through oxidative stress and excitotoxicity. Long-term users show reduced dopamine transporter density in striatum and prefrontal cortex. Some recovery occurs with abstinence, but cognitive deficits (particularly in executive function and memory) may persist for years. The damage correlates with cumulative dose and duration of use.
Glutamate receptor overstimulation causes excitotoxicity—neurons literally excited to death. Excessive glutamate triggers massive calcium influx, activating proteases and nucleases that destroy cellular components, ultimately triggering apoptosis. PCP and ketamine act as glutamate agonists at high doses, potentially causing neuronal death. Even marijuana contains 400+ chemicals, making it difficult to isolate which might be harmful. THC itself appears relatively safe, but combustion products (when smoked) include carcinogens and toxins.
Treatment: From Abstinence to Harm Reduction
Treatment approaches range from complete abstinence (12-step programs) to harm reduction (reducing negative consequences without requiring abstinence). Medication-assisted treatment uses pharmacology to manage addiction. Buprenorphine and methadone are long-acting opioid agonists that prevent withdrawal and craving while blocking effects of short-acting opioids. They allow addicts to stabilize their lives without perpetual drug-seeking.
Naltrexone, an opioid antagonist, blocks opioid effects entirely, used after detoxification to prevent relapse. For alcohol, naltrexone reduces the reinforcing effects of drinking, helping patients reduce consumption. Disulfiram blocks aldehyde dehydrogenase, causing toxic acetaldehyde accumulation if alcohol is consumed—people become violently ill if they drink. The fear of this reaction can support abstinence.
Cognitive behavioral therapy teaches coping strategies, identifies triggers, and develops relapse prevention plans. Contingency management provides tangible rewards for abstinence, leveraging our understanding of reward circuits. The most effective treatments combine medication, therapy, and social support, recognizing that addiction involves biological, psychological, and social factors.
Conclusion: The Chemistry of Choice and Change
Psychopharmacology reveals a profound truth: our thoughts, emotions, and behaviors are chemical processes that can be modified by molecules. This doesn't reduce human experience to mere chemistry—love, creativity, and consciousness emerge from these processes but aren't identical to them. Understanding the mechanisms empowers us to intervene rationally. We can treat schizophrenia by blocking dopamine receptors, depression by enhancing monoamines, addiction by targeting reward circuits.
But this power brings responsibility. Every psychoactive substance—therapeutic or recreational—alters the delicate balance of neurotransmitter systems shaped by millions of years of evolution. The same drugs that relieve suffering can cause dependence, impair cognition, or damage neurons. The challenge is maximizing benefits while minimizing harms—understanding pharmacology, respecting individual differences, recognizing the role of set and setting.
The distinction between drugs and behaviors blurs when we understand mechanism. Video games, sugar, and social media engage the same circuits as cocaine or heroin, just with different magnitudes. This doesn't mean they're equally dangerous, but it does mean we should apply the same framework: Is the behavior producing benefits or causing harm? Is control maintained or lost? Are there better ways to activate reward circuits—through exercise, social connection, meaningful work, or creative expression?
Perhaps most fundamentally, psychopharmacology demonstrates the material basis of consciousness. Your subjective experience—the felt quality of being you—depends on the precise concentrations of dozens of neurotransmitters acting on hundreds of receptor subtypes in billions of synapses. Shift serotonin levels, and your mood changes. Block dopamine receptors, and reality perception alters. Activate 5-HT2A receptors, and the boundaries of self dissolve. The fact that molecules can so reliably modify consciousness argues that consciousness is what brains do, not something separate that brains generate.
Thought Questions for Discussion
The Authenticity Question: If antidepressants restore normal mood by increasing serotonin, is the resulting happiness "authentic"? If psychedelics produce mystical experiences through 5-HT2A activation, are these experiences genuine spiritual insights or just chemical illusions? What does it mean for an experience to be "real" if all experiences have chemical correlates? Does the mechanism of production affect the validity of the experience?
The Addiction Paradox: If addiction involves dopamine hijacking reward circuits and prefrontal dysfunction impairing control, to what extent are addicts responsible for their behavior? Does understanding the neuroscience reduce moral culpability? Yet if we remove all responsibility, how do we motivate recovery? How do we balance compassion based on neurobiological understanding with accountability necessary for treatment?
The Enhancement Dilemma: We accept drugs that restore function (antidepressants for depression, stimulants for ADHD), but debate drugs that enhance normal function (modafinil for focus, memory-enhancing drugs for studying). Where is the line between treatment and enhancement? Should we allow cognitive enhancement drugs if they're safe? What about mandatory enhancement in competitive environments (military, academics, athletics)? Does neuroscience give us tools to improve ourselves beyond natural variation, and should we use them?
Practice Questions:
• The blood-brain barrier is formed by _______ junctions between capillary endothelial cells, with _______ end feet covering 80% of the capillary surface.
• Drugs can modify synaptic transmission at seven points: synthesis, storage, _______, receptor binding, _______, enzymatic degradation, and second messengers.
• Benzodiazepines act as _______ at GABA-A receptors, increasing the _______ of chloride channel opening.
• All effective antipsychotics share the property of blocking _______ receptors, particularly the _______ subtype.
• SSRIs work by blocking the _______ transporter, increasing synaptic serotonin levels.
• According to incentive-sensitization theory, addiction involves dissociation between _______ (mediated by opioids) and _______ (mediated by dopamine).
• All addictive drugs increase _______ in the nucleus accumbens, either directly or indirectly.
• With repeated drug use, control shifts from _______ striatum (goal-directed) to _______ striatum (habitual).
📝 View Answer Key for Chapter 13